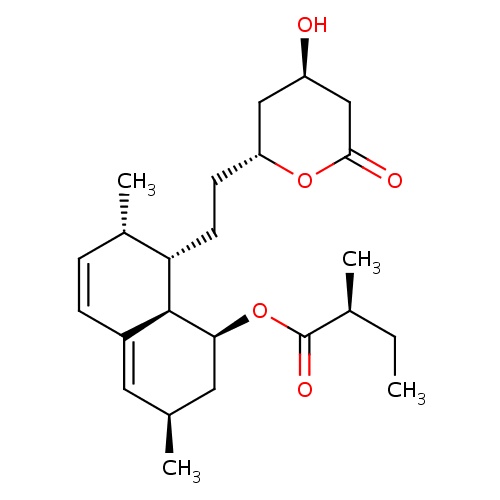
IUPAC name
(1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (2S)-2-methylbutanoate
SMILES
[H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H]1C[C@@H](O)CC(=O)O1)OC(=O)[C@@H](C)CC
Compound class
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
For management as an adjunct to diet to reduce elevated total-C, LDL-C, apo B, and TG levels in patients with primary hypercholesterolemia and mixed dyslipidemia. For primary prevention of coronary heart disease and to slow progression of coronary atherosclerosis in patients with coronary heart disease.
Common name
Lovastatin
IUPAC name
(1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (2S)-2-methylbutanoate
SMILES
[H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H]1C[C@@H](O)CC(=O)O1)OC(=O)[C@@H](C)CC
INCHI
InChI=1S/C24H36O5/c1-5-15(3)24(27)29-21-11-14(2)10-17-7-6-16(4)20(23(17)21)9-8-19-12-18(25)13-22(26)28-19/h6-7,10,14-16,18-21,23,25H,5,8-9,11-13H2,1-4H3/t14-,15-,16-,18+,19+,20-,21-,23-/m0/s1
FORMULA
C24H36O5

Common name
Lovastatin
IUPAC name
(1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (2S)-2-methylbutanoate
Molecular weight
404.540
clogP
3.513
clogS
-3.187
HBond Acceptor
5
HBond Donor
1
Total Polar Surface Area
72.83
Number of Rings
3
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
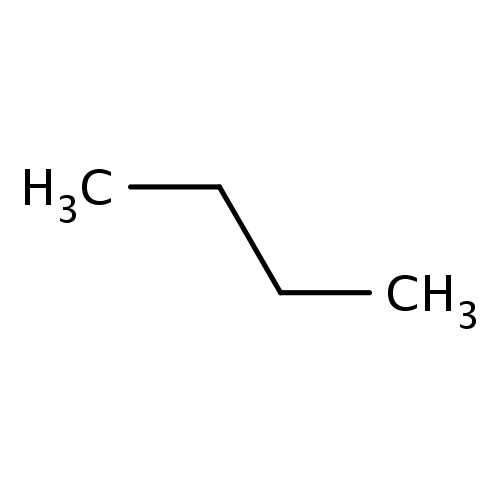
| FDBF00067 | butane |
|
CCCC | 0.0680 |
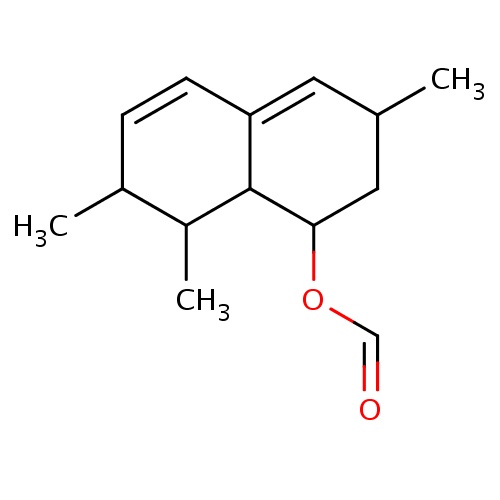
| FDBF00455 | [(1S,3R,7S,8S,8aR)-3,7,8-trimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] formate |
|
CC1C2C(=CC(CC2OC=O)C)C=CC1C | 0.0007 |
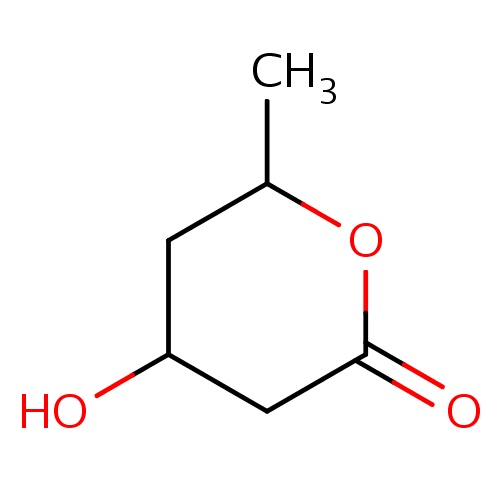
| FDBF00456 | (4R,6R)-4-hydroxy-6-methyl-tetrahydropyran-2-one |
|
CC1OC(=O)CC(C1)O | 0.0007 |
| FDBF00457 | [(1S,3R,7R,8aR)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] propanoate |
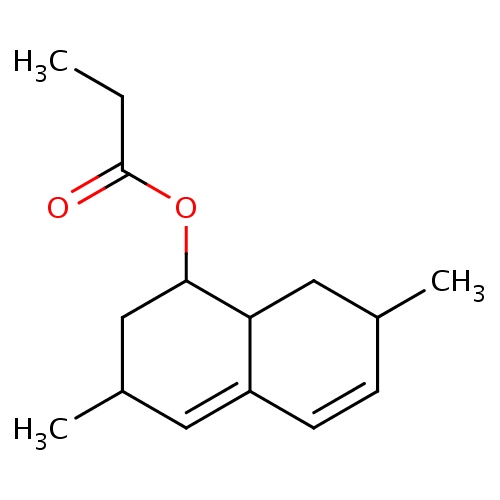
|
O(C(=O)CC)C1C2CC(C=CC2=CC(C1)C)C | 0.0003 |
| FDBF00458 | [(1S,3R,7S,8S,8aR)-8-ethyl-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] formate |

|
CCC1C2C(=CC(CC2OC=O)C)C=CC1C | 0.0007 |
| FDBF00459 | [(1S,3R,7S,8S,8aR)-3,7,8-trimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] propanoate |
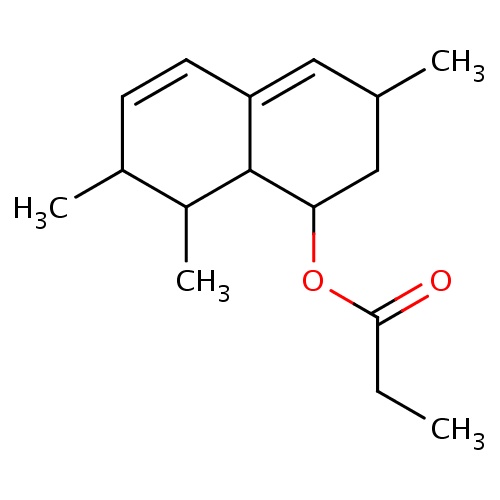
|
CC1C2C(=CC(CC2OC(=O)CC)C)C=CC1C | 0.0003 |
| FDBF00460 | (4R,6R)-6-ethyl-4-hydroxy-tetrahydropyran-2-one |
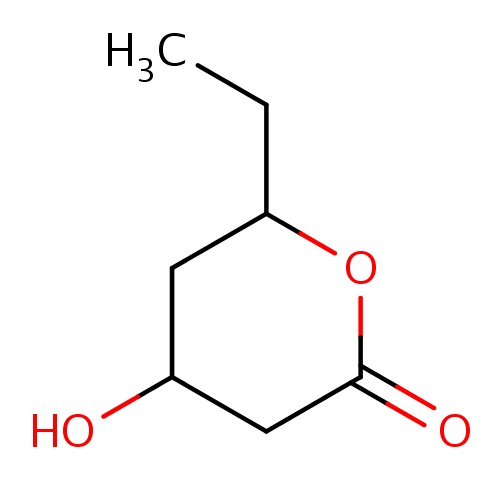
|
C(C)C1OC(=O)CC(C1)O | 0.0007 |
| FDBF00461 | [(1S,3R,7R,8aR)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] formate |

|
O(C=O)C1C2CC(C=CC2=CC(C1)C)C | 0.0007 |
| FDBF00462 | (4R)-4-hydroxytetrahydropyran-2-one |
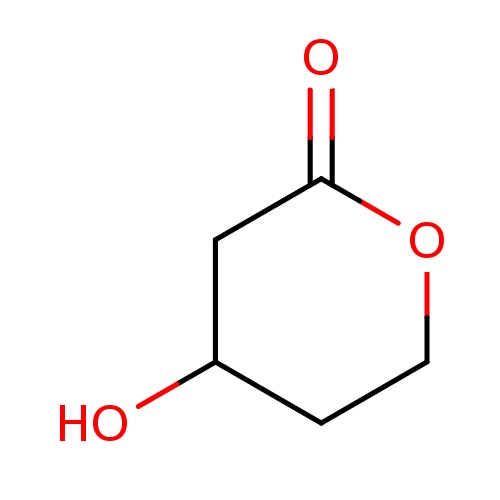
|
O1CCC(CC1=O)O | 0.0007 |

