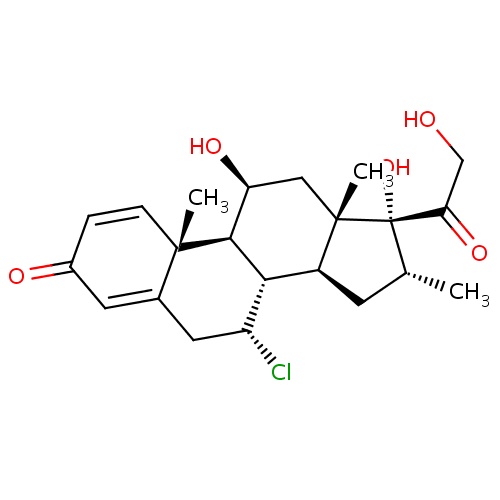
IUPAC name
(1S,2R,9R,10S,11S,13R,14R,15S,17S)-9-chloro-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,13,15-trimethyltetracyclo[8.7.0.0²,
SMILES
[H][C@@]12C[C@@H](C)[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1([H])[C@@]2([H])[C@H](Cl)CC2=CC(=O)C=C[C@]12C
Compound class
Antipruritics; Corticosteroids; Ophthalmologicals; Sensory Organs; Dermatologicals; Corticosteroids, Plain; Corticosteroids, Dermatological Preparations; Corticosteroids, Moderately Potent (Group II); Antiinflammatory Agents; CYP3A4 Inhibitors;
Therapeutic area
For the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Common name
Alclometasone
IUPAC name
(1S,2R,9R,10S,11S,13R,14R,15S,17S)-9-chloro-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,13,15-trimethyltetracyclo[8.7.0.0²,
SMILES
[H][C@@]12C[C@@H](C)[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1([H])[C@@]2([H])[C@H](Cl)CC2=CC(=O)C=C[C@]12C
INCHI
InChI=1S/C22H29ClO5/c1-11-6-14-18-15(23)8-12-7-13(25)4-5-20(12,2)19(18)16(26)9-21(14,3)22(11,28)17(27)10-24/h4-5,7,11,14-16,18-19,24,26,28H,6,8-10H2,1-3H3/t11-,14+,15-,16+,18-,19+,20+,21+,22+/m1/s1
FORMULA
C22H29ClO5

Common name
Alclometasone
IUPAC name
(1S,2R,9R,10S,11S,13R,14R,15S,17S)-9-chloro-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,13,15-trimethyltetracyclo[8.7.0.0²,
Molecular weight
408.916
clogP
2.406
clogS
-2.670
HBond Acceptor
5
HBond Donor
3
Total Polar Surface Area
94.83
Number of Rings
4
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
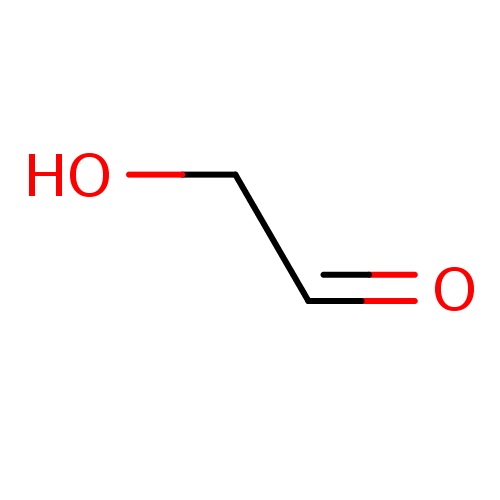
| FDBF00204 | 2-hydroxyacetaldehyde |
|
O=CCO | 0.0103 |

