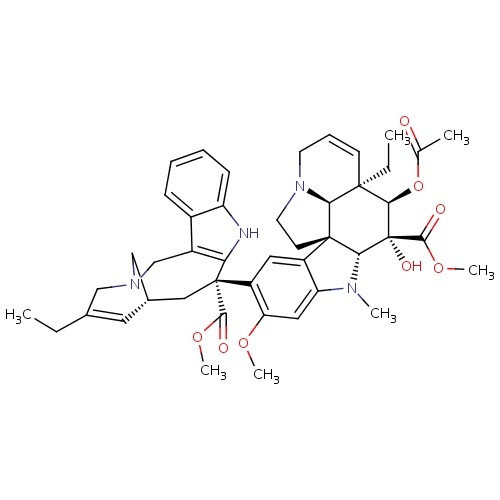
IUPAC name
methyl (1R,9R,10R,11R,12R,19R)-11-(acetyloxy)-12-ethyl-4-[(12S,14R)-16-ethyl-12-(methoxycarbonyl)-1,10-diazatetracyclo[12.3.1.0³,¹¹.0
SMILES
[H][C@@]12N(C)C3=CC(OC)=C(C=C3[C@@]11CCN3CC=C[C@@](CC)([C@@H](OC(C)=O)[C@@]2(O)C(=O)OC)[C@@]13[H])[C@]1(C[C@@]2([H])CN(CC(CC)=C2)CC2=C1NC1=CC=CC=C21)C(=O)OC
Compound class
Antineoplastic Agents; Immunosuppressive Agents; Antineoplastic Agents, Phytogenic; Antineoplastic and Immunomodulating Agents; Vinca Alkaloids and Analogues; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
For the treatment of non-small-cell lung carcinoma.
Common name
Vinorelbine
IUPAC name
methyl (1R,9R,10R,11R,12R,19R)-11-(acetyloxy)-12-ethyl-4-[(12S,14R)-16-ethyl-12-(methoxycarbonyl)-1,10-diazatetracyclo[12.3.1.0³,¹¹.0
SMILES
[H][C@@]12N(C)C3=CC(OC)=C(C=C3[C@@]11CCN3CC=C[C@@](CC)([C@@H](OC(C)=O)[C@@]2(O)C(=O)OC)[C@@]13[H])[C@]1(C[C@@]2([H])CN(CC(CC)=C2)CC2=C1NC1=CC=CC=C21)C(=O)OC
INCHI
InChI=1S/C45H54N4O8/c1-8-27-19-28-22-44(40(51)55-6,36-30(25-48(23-27)24-28)29-13-10-11-14-33(29)46-36)32-20-31-34(21-35(32)54-5)47(4)38-43(31)16-18-49-17-12-15-42(9-2,37(43)49)39(57-26(3)50)45(38,53)41(52)56-7/h10-15,19-21,28,37-39,46,53H,8-9,16-18,22-25H2,1-7H3/t28-,37-,38+,39+,42+,43+,44-,45+/m0/s1
FORMULA
C45H54N4O8

Common name
Vinorelbine
IUPAC name
methyl (1R,9R,10R,11R,12R,19R)-11-(acetyloxy)-12-ethyl-4-[(12S,14R)-16-ethyl-12-(methoxycarbonyl)-1,10-diazatetracyclo[12.3.1.0³,¹¹.0
Molecular weight
778.932
clogP
4.965
clogS
-7.664
HBond Acceptor
11
HBond Donor
2
Total Polar Surface Area
133.87
Number of Rings
9
Rotatable Bond
10