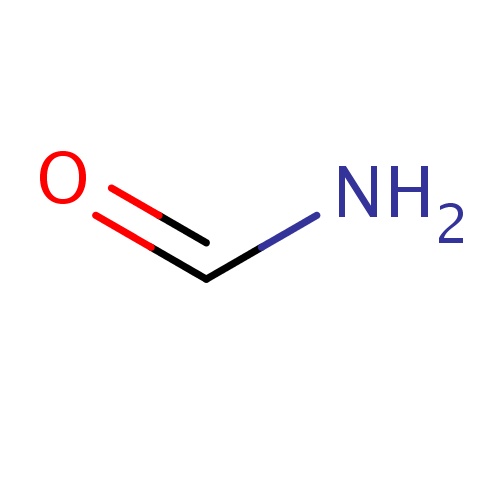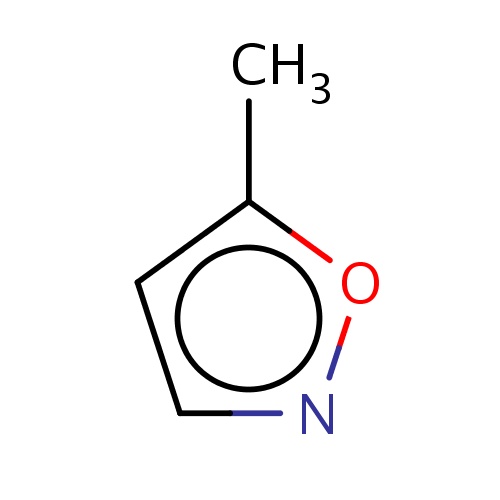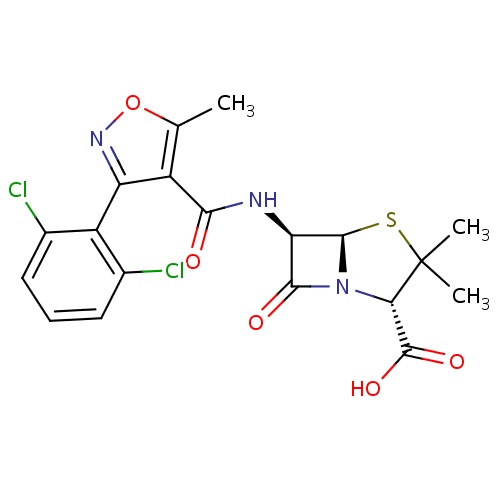
IUPAC name
(2S,5R,6R)-6-[3-(2,6-dichlorophenyl)-5-methyl-1,2-oxazole-4-amido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
SMILES
[H][C@]12SC(C)(C)[C@@H](N1C(=O)[C@H]2NC(=O)C1=C(C)ON=C1C1=C(Cl)C=CC=C1Cl)C(O)=O
Compound class
Anti-Bacterial Agents; Penicillins; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Antibacterials for Intramammary Use; Beta-Lactamase Resistant Penicillins; Beta-Lactam Antibacterials, Penicillins; Beta-Lactam Antibacterials, Penicillins, for Intramammary Use; CYP3A4 Inhibitors;
Therapeutic area
Used to treat infections caused by penicillinase-producing staphylococci which have demonstrated susceptibility to the drug.
Common name
Dicloxacillin
IUPAC name
(2S,5R,6R)-6-[3-(2,6-dichlorophenyl)-5-methyl-1,2-oxazole-4-amido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
SMILES
[H][C@]12SC(C)(C)[C@@H](N1C(=O)[C@H]2NC(=O)C1=C(C)ON=C1C1=C(Cl)C=CC=C1Cl)C(O)=O
INCHI
InChI=1S/C19H17Cl2N3O5S/c1-7-10(12(23-29-7)11-8(20)5-4-6-9(11)21)15(25)22-13-16(26)24-14(18(27)28)19(2,3)30-17(13)24/h4-6,13-14,17H,1-3H3,(H,22,25)(H,27,28)/t13-,14+,17-/m1/s1
FORMULA
C19H17Cl2N3O5S

Common name
Dicloxacillin
IUPAC name
(2S,5R,6R)-6-[3-(2,6-dichlorophenyl)-5-methyl-1,2-oxazole-4-amido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
Molecular weight
470.326
clogP
2.887
clogS
-4.938
HBond Acceptor
6
HBond Donor
2
Total Polar Surface Area
138.04
Number of Rings
4
Rotatable Bond
4