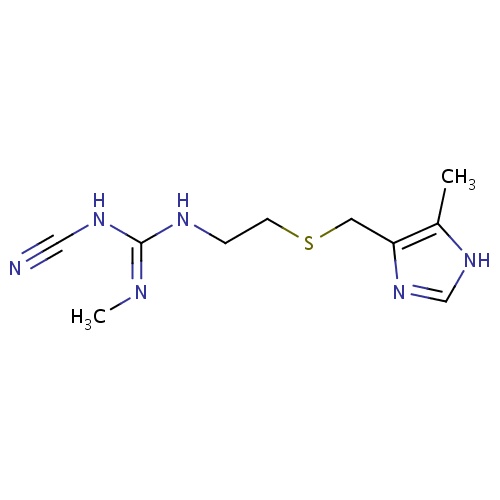
IUPAC name
(Z)-1-cyano-2-methyl-3-(2-{[(5-methyl-1H-imidazol-4-yl)methyl]sulfanyl}ethyl)guanidine
SMILES
C\N=C(\NCCSCC1=C(C)NC=N1)NC#N
Compound class
Anti-Ulcer Agents; Adjuvants; Alimentary Tract and Metabolism; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; H2 Antagonists; BSEP/ABCB11 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
For the treatment and the management of acid-reflux disorders (GERD), peptic ulcer disease, heartburn, and acid indigestion.
Common name
Cimetidine
IUPAC name
(Z)-1-cyano-2-methyl-3-(2-{[(5-methyl-1H-imidazol-4-yl)methyl]sulfanyl}ethyl)guanidine
SMILES
C\N=C(\NCCSCC1=C(C)NC=N1)NC#N
INCHI
InChI=1S/C10H16N6S/c1-8-9(16-7-15-8)5-17-4-3-13-10(12-2)14-6-11/h7H,3-5H2,1-2H3,(H,15,16)(H2,12,13,14)
FORMULA
C10H16N6S

Common name
Cimetidine
IUPAC name
(Z)-1-cyano-2-methyl-3-(2-{[(5-methyl-1H-imidazol-4-yl)methyl]sulfanyl}ethyl)guanidine
Molecular weight
252.339
clogP
1.519
clogS
-3.471
HBond Acceptor
3
HBond Donor
3
Total Polar Surface Area
114.19
Number of Rings
1
Rotatable Bond
8
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
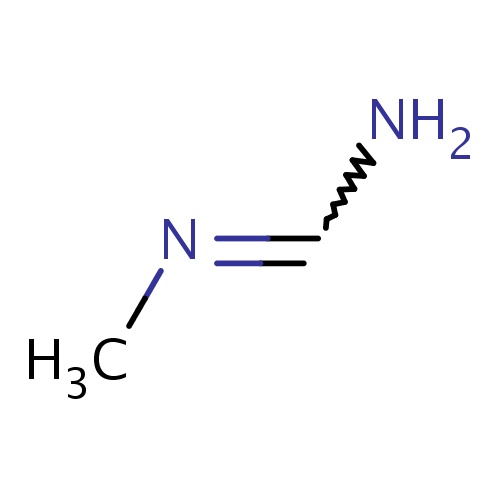
| FDBF00389 | N'-methylformamidine |
|
NC=NC | 0.0024 |

