
IUPAC name
1,3-thiazol-5-ylmethyl N-[(2S,3S,5S)-3-hydroxy-5-[(2S)-3-methyl-2-{[methyl({[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl})carbamoyl]amino}butanamido]-1,6-diphenylhexan-2-yl]carbamate
SMILES
CC(C)[C@H](NC(=O)N(C)CC1=CSC(=N1)C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC1=CC=CC=C1)NC(=O)OCC1=CN=CS1)CC1=CC=CC=C1
Compound class
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
Indicated in combination with other antiretroviral agents for the treatment of HIV-infection.
Common name
Ritonavir
IUPAC name
1,3-thiazol-5-ylmethyl N-[(2S,3S,5S)-3-hydroxy-5-[(2S)-3-methyl-2-{[methyl({[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl})carbamoyl]amino}butanamido]-1,6-diphenylhexan-2-yl]carbamate
SMILES
CC(C)[C@H](NC(=O)N(C)CC1=CSC(=N1)C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC1=CC=CC=C1)NC(=O)OCC1=CN=CS1)CC1=CC=CC=C1
INCHI
InChI=1S/C37H48N6O5S2/c1-24(2)33(42-36(46)43(5)20-29-22-49-35(40-29)25(3)4)34(45)39-28(16-26-12-8-6-9-13-26)18-32(44)31(17-27-14-10-7-11-15-27)41-37(47)48-21-30-19-38-23-50-30/h6-15,19,22-25,28,31-33,44H,16-18,20-21H2,1-5H3,(H,39,45)(H,41,47)(H,42,46)/t28-,31-,32-,33-/m0/s1
FORMULA
C37H48N6O5S2

Common name
Ritonavir
IUPAC name
1,3-thiazol-5-ylmethyl N-[(2S,3S,5S)-3-hydroxy-5-[(2S)-3-methyl-2-{[methyl({[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl})carbamoyl]amino}butanamido]-1,6-diphenylhexan-2-yl]carbamate
Molecular weight
720.944
clogP
7.333
clogS
-8.053
HBond Acceptor
7
HBond Donor
4
Total Polar Surface Area
202.26
Number of Rings
4
Rotatable Bond
19
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00023 | toluene |
|
c1(ccccc1)C | 0.1268 |
| FDBF00042 | propan-2-ol |
|
CC(O)C | 0.0278 |
| FDBF00141 | ethylbenzene |
|
c1(ccccc1)CC | 0.0371 |
| FDBF00272 | pentan-3-ol |
|
OC(CC)CC | 0.0017 |
| FDBF00416 | butan-1-ol |
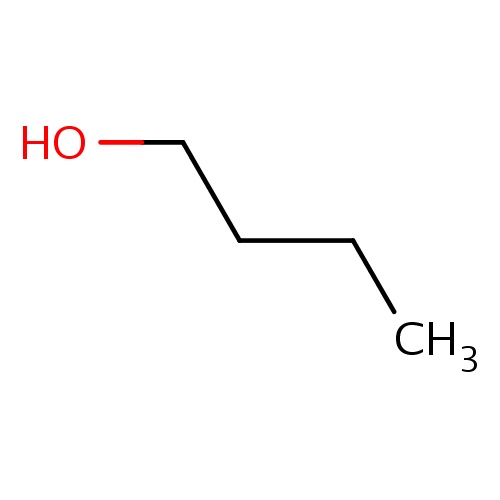
|
C(CC)CO | 0.0134 |
| FDBF01127 | methylcarbamic acid |

|
O=C(O)NC | 0.0199 |
| FDBF01143 | N-isopropylacetamide |

|
CC(C)NC(=O)C | 0.0024 |
| FDBF01146 | N-ethyl-3-methyl-butanamide |
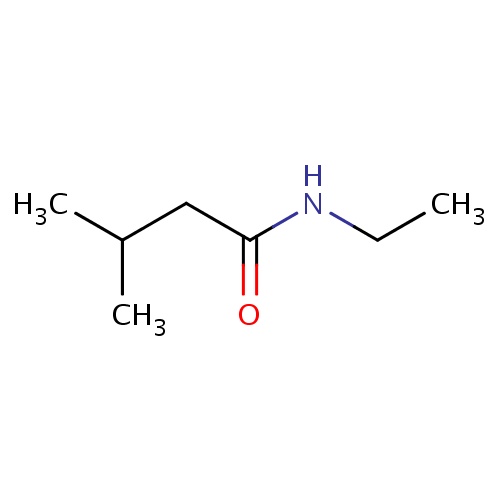
|
C(C)(C)CC(=O)NCC | 0.0010 |
| FDBF01153 | N-[(1S)-3-hydroxy-1-methyl-propyl]acetamide |
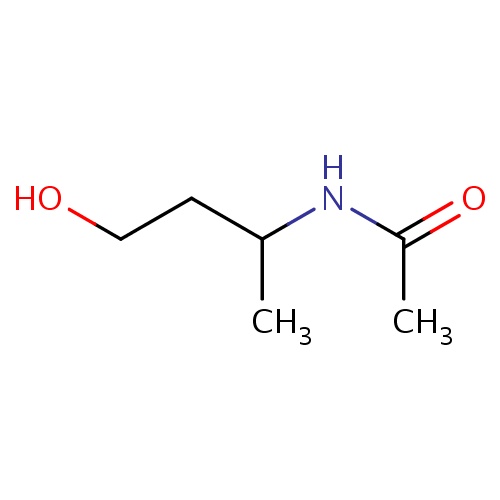
|
CC(CCO)NC(=O)C | 0.0003 |
| FDBF01155 | N-isopropyl-3-methyl-butanamide |
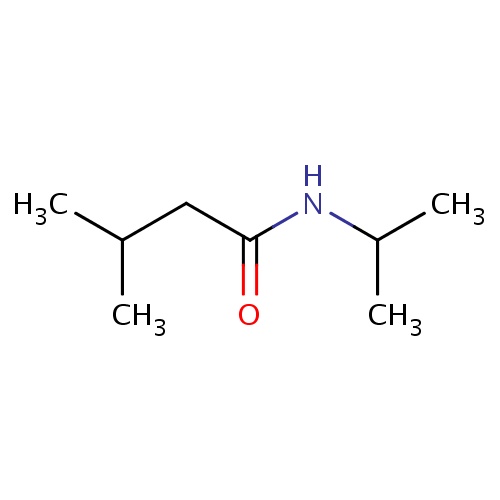
|
C(C)(C)CC(=O)NC(C)C | 0.0007 |

