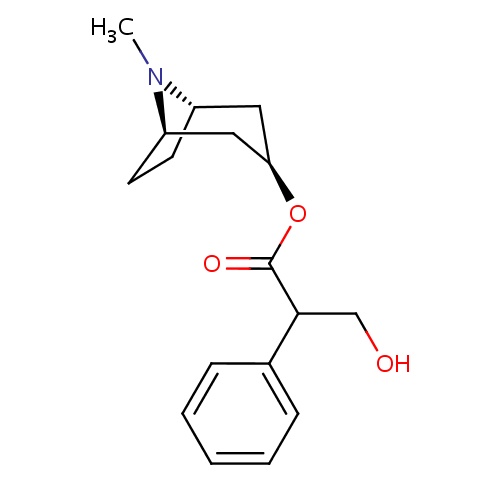
IUPAC name
(1R,3R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl 3-hydroxy-2-phenylpropanoate
SMILES
CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(=O)C(CO)C1=CC=CC=C1
Compound class
Anti-Arrhythmia Agents; Bronchodilator Agents; Muscarinic Antagonists; Parasympatholytics; Adjuvants, Anesthesia; Mydriatics; Ophthalmologicals; Sensory Organs; Alimentary Tract and Metabolism; Drugs for Functional Gastrointestinal Disorders; Anticholinergics; Mydriatics and Cycloplegics; Belladonna Alkaloids, Tertiary Amines; Belladonna and Derivatives, Plain; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers;
Therapeutic area
For the treatment of poisoning by susceptible organophosphorous nerve agents having anti-cholinesterase activity (cholinesterase inhibitors) as well as organophosphorous or carbamate insecticides.
Common name
Atropine
IUPAC name
(1R,3R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl 3-hydroxy-2-phenylpropanoate
SMILES
CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(=O)C(CO)C1=CC=CC=C1
INCHI
InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3/t13-,14+,15+,16?
FORMULA
C17H23NO3

Common name
Atropine
IUPAC name
(1R,3R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl 3-hydroxy-2-phenylpropanoate
Molecular weight
289.369
clogP
2.033
clogS
-2.173
HBond Acceptor
4
HBond Donor
1
Total Polar Surface Area
49.77
Number of Rings
3
Rotatable Bond
5
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
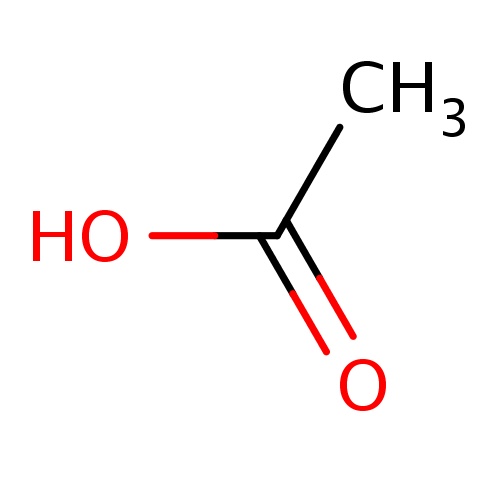
| FDBF00004 | acetic acid |
|
CC(=O)O | 0.0687 |
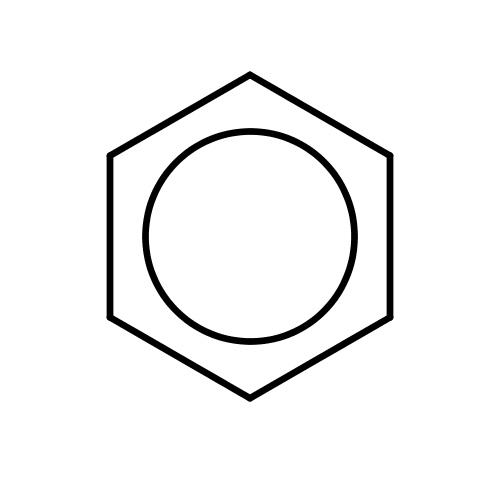
| FDBF00005 | benzene |
|
c1ccccc1 | 0.2824 |
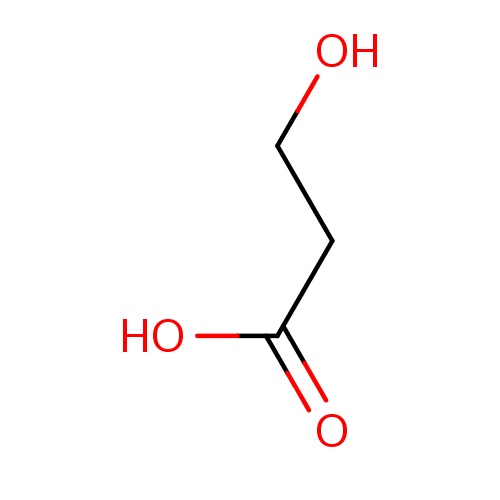
| FDBF00149 | 3-hydroxypropanoic acid |
|
C(O)CC(=O)O | 0.0038 |
| FDBF00506 | (1S,5R)-8-methyl-8-azabicyclo[3.2.1]octane |

|
C12CCCC(N1C)CC2 | 0.0024 |
| FDBF00731 | 2-phenylacetic acid |

|
OC(=O)Cc1ccccc1 | 0.0024 |
| FDBF00734 | (2S)-3-hydroxy-2-phenyl-propanoic acid |

|
C(O)C(C(=O)O)c1ccccc1 | 0.0010 |
| FDBF00919 | [(1R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl] acetate |

|
O(C(=O)C)C1CC2CCC(C1)N2C | 0.0007 |

