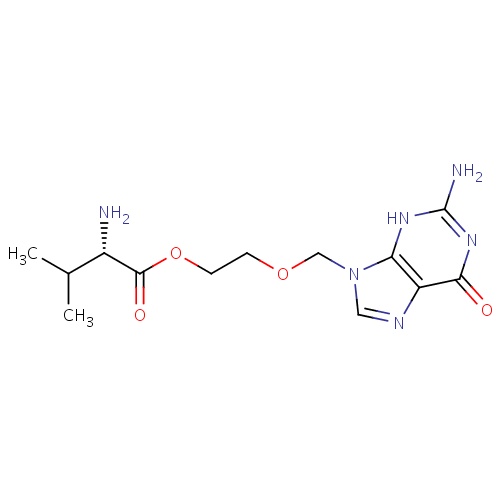
IUPAC name
2-[(2-amino-6-oxo-6,9-dihydro-3H-purin-9-yl)methoxy]ethyl (2S)-2-amino-3-methylbutanoate
SMILES
CC(C)[C@H](N)C(=O)OCCOCN1C=NC2=C1NC(N)=NC2=O
Compound class
Antiviral Agents; Prodrugs; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Nucleosides and Nucleotides Excl. Reverse Transcriptase Inhibitors;
Therapeutic area
For the treatment or suppression of cold sores (herpes labialis), herpes zoster (shingles), genital herpes in immunocompetent individuals, and recurrent genital herpes in HIV-infected individuals.
Common name
Valaciclovir
IUPAC name
2-[(2-amino-6-oxo-6,9-dihydro-3H-purin-9-yl)methoxy]ethyl (2S)-2-amino-3-methylbutanoate
SMILES
CC(C)[C@H](N)C(=O)OCCOCN1C=NC2=C1NC(N)=NC2=O
INCHI
InChI=1S/C13H20N6O4/c1-7(2)8(14)12(21)23-4-3-22-6-19-5-16-9-10(19)17-13(15)18-11(9)20/h5,7-8H,3-4,6,14H2,1-2H3,(H3,15,17,18,20)/t8-/m0/s1
FORMULA
C13H20N6O4

Common name
Valaciclovir
IUPAC name
2-[(2-amino-6-oxo-6,9-dihydro-3H-purin-9-yl)methoxy]ethyl (2S)-2-amino-3-methylbutanoate
Molecular weight
324.336
clogP
-0.240
clogS
-1.872
HBond Acceptor
6
HBond Donor
5
Total Polar Surface Area
151.14
Number of Rings
2
Rotatable Bond
8
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00007 | propane |

|
C(C)C | 0.2412 |
| FDBF00100 | ethyl formate |

|
O(C=O)CC | 0.0244 |
| FDBF00105 | methyl formate |
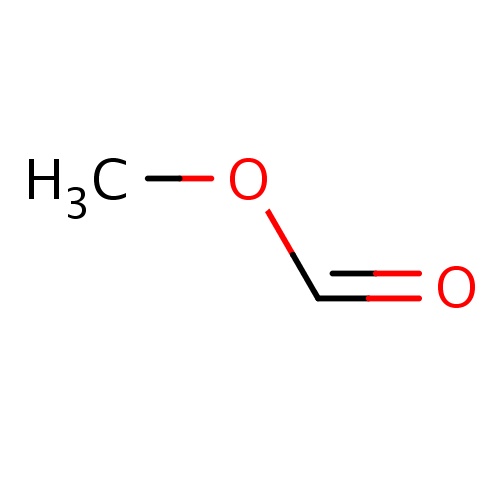
|
O(C=O)C | 0.0323 |
| FDBF00158 | methoxymethane |
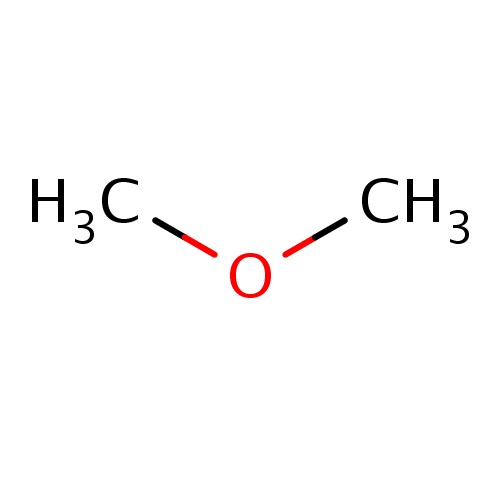
|
COC | 0.0374 |
| FDBF00191 | ethyl 2-aminoacetate |
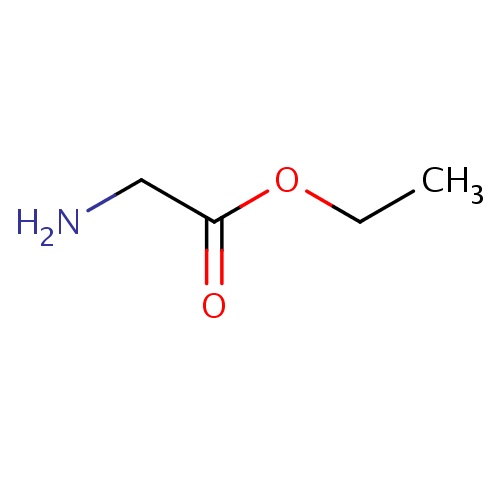
|
C(N)C(=O)OCC | 0.0041 |
| FDBF01296 | 2-methylpropan-1-amine |
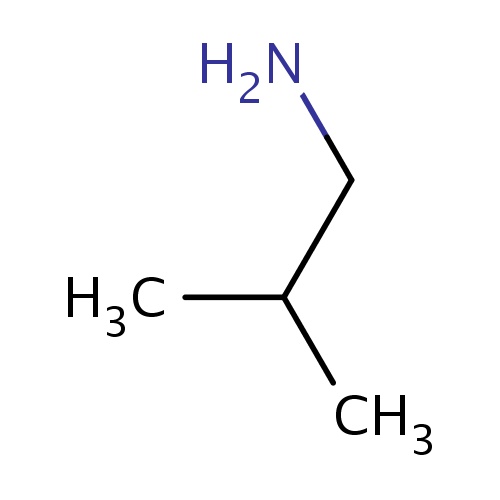
|
NCC(C)C | 0.0017 |
| FDBF01297 | methyl 2-aminoacetate |

|
NCC(=O)OC | 0.0010 |
| FDBF01298 | methyl (2S)-2-amino-3-methyl-butanoate |
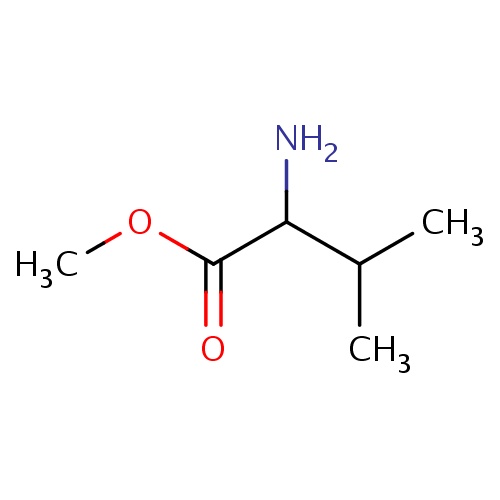
|
NC(C(=O)OC)C(C)C | 0.0007 |
| FDBF01299 | 2-hydroxyethyl formate |
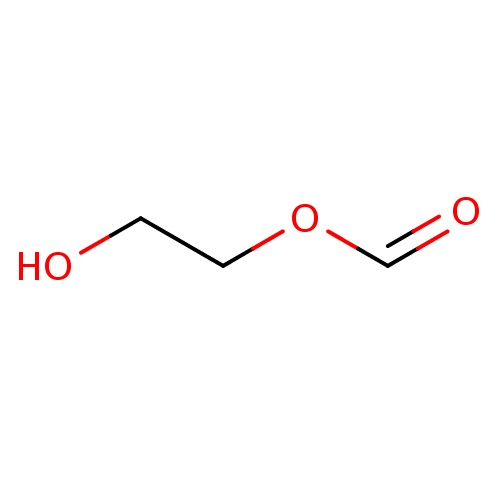
|
OCCOC=O | 0.0017 |

