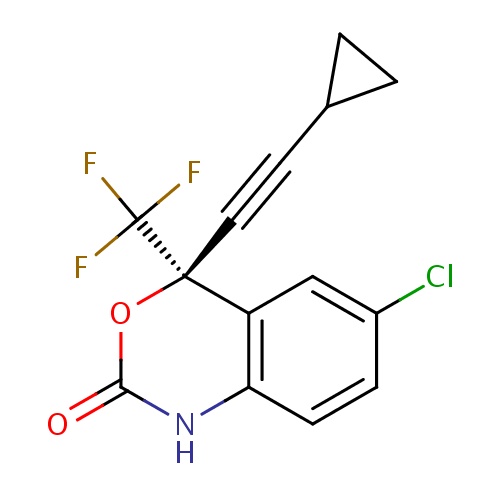
IUPAC name
(4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-2,4-dihydro-1H-3,1-benzoxazin-2-one
SMILES
FC(F)(F)[C@]1(OC(=O)NC2=C1C=C(Cl)C=C2)C#CC1CC1
Compound class
Anti-HIV Agents; Reverse Transcriptase Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Non-Nucleoside Reverse Transcriptase Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP3A Inducers; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
For use in combination treatment of HIV infection (AIDS).
Common name
Efavirenz
IUPAC name
(4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-2,4-dihydro-1H-3,1-benzoxazin-2-one
SMILES
FC(F)(F)[C@]1(OC(=O)NC2=C1C=C(Cl)C=C2)C#CC1CC1
INCHI
InChI=1S/C14H9ClF3NO2/c15-9-3-4-11-10(7-9)13(14(16,17)18,21-12(20)19-11)6-5-8-1-2-8/h3-4,7-8H,1-2H2,(H,19,20)/t13-/m0/s1
FORMULA
C14H9ClF3NO2

Common name
Efavirenz
IUPAC name
(4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-2,4-dihydro-1H-3,1-benzoxazin-2-one
Molecular weight
315.675
clogP
4.012
clogS
-4.330
HBond Acceptor
2
HBond Donor
1
Total Polar Surface Area
38.33
Number of Rings
3
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
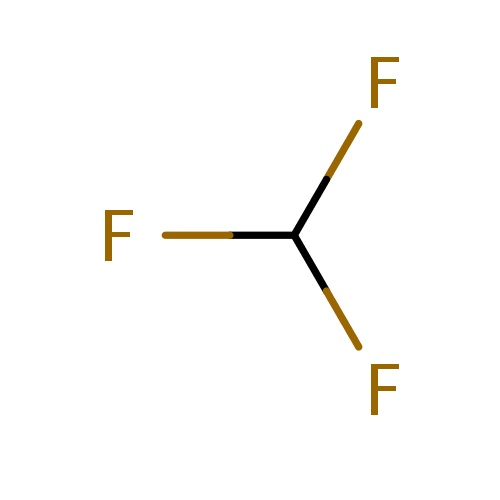
| FDBF00177 | fluoroform |
|
FC(F)F | 0.0704 |
| FDBF00259 | cyclopropane |

|
C1CC1 | 0.0175 |
| FDBF01429 | ethynylcyclopropane |

|
C(#C)C1CC1 | 0.0003 |
| FDBF01430 | (4S)-6-chloro-4-ethynyl-1,4-dihydro-3,1-benzoxazin-2-one |

|
Clc1cc2c(cc1)NC(=O)OC2C#C | 0.0003 |
| FDBF01431 | (4S)-6-chloro-4-(trifluoromethyl)-1,4-dihydro-3,1-benzoxazin-2-one |

|
FC(F)(F)C1c2cc(ccc2NC(=O)O1)Cl | 0.0003 |
| FDBF01432 | 6-chloro-1,4-dihydro-3,1-benzoxazin-2-one |

|
Clc1cc2c(cc1)NC(=O)OC2 | 0.0003 |

