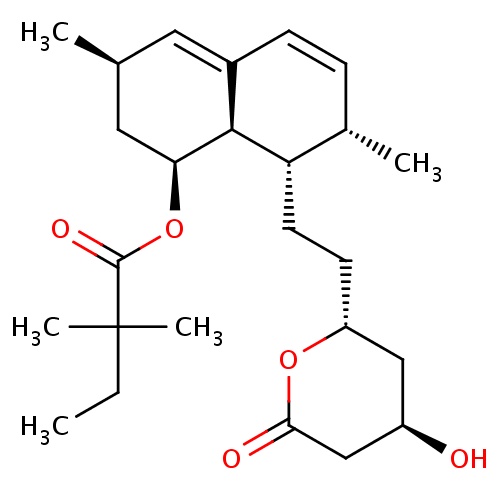
IUPAC name
(1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl 2,2-dimethylbutanoate
SMILES
[H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H]1C[C@@H](O)CC(=O)O1)OC(=O)C(C)(C)CC
Compound class
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; Hypolipidemic Agents; Drugs Used in Diabetes; Alimentary Tract and Metabolism; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Blood Glucose Lowering Drugs, Excl. Insulins; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
For the treatment of hypercholesterolemia and for the reduction in the risk of cardiac heart disease mortality and cardiovascular events. It can also be used in adolescent patients for the treatment of heterozygous familial hypercholesterolemia.
Common name
Simvastatin
IUPAC name
(1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl 2,2-dimethylbutanoate
SMILES
[H][C@]12[C@H](C[C@@H](C)C=C1C=C[C@H](C)[C@@H]2CC[C@@H]1C[C@@H](O)CC(=O)O1)OC(=O)C(C)(C)CC
INCHI
InChI=1S/C25H38O5/c1-6-25(4,5)24(28)30-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-19-13-18(26)14-22(27)29-19/h7-8,11,15-16,18-21,23,26H,6,9-10,12-14H2,1-5H3/t15-,16-,18+,19+,20-,21-,23-/m0/s1
FORMULA
C25H38O5

Common name
Simvastatin
IUPAC name
(1S,3R,7S,8S,8aR)-8-{2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl}-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl 2,2-dimethylbutanoate
Molecular weight
418.566
clogP
3.765
clogS
-3.562
HBond Acceptor
5
HBond Donor
1
Total Polar Surface Area
72.83
Number of Rings
3
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00007 | propane |

|
C(C)C | 0.2412 |
| FDBF00115 | 2-methylbutane |

|
C(C)(C)CC | 0.0103 |
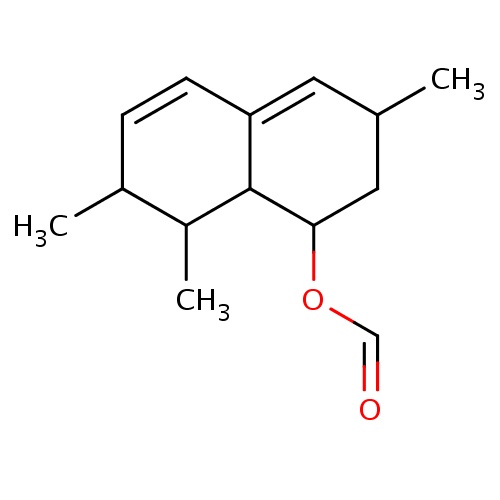
| FDBF00455 | [(1S,3R,7S,8S,8aR)-3,7,8-trimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] formate |
|
CC1C2C(=CC(CC2OC=O)C)C=CC1C | 0.0007 |
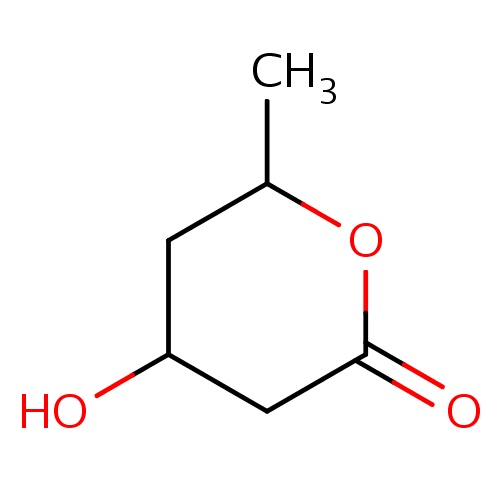
| FDBF00456 | (4R,6R)-4-hydroxy-6-methyl-tetrahydropyran-2-one |
|
CC1OC(=O)CC(C1)O | 0.0007 |
| FDBF00458 | [(1S,3R,7S,8S,8aR)-8-ethyl-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] formate |

|
CCC1C2C(=CC(CC2OC=O)C)C=CC1C | 0.0007 |
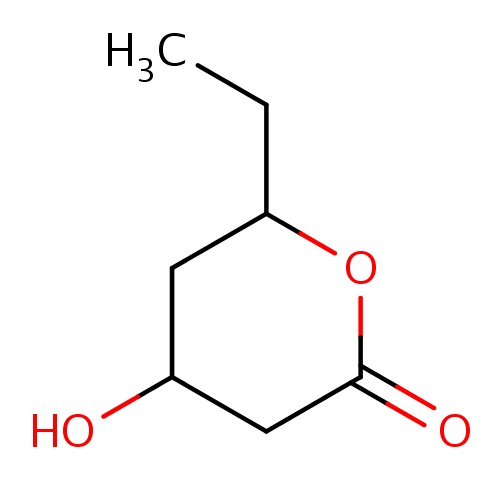
| FDBF00460 | (4R,6R)-6-ethyl-4-hydroxy-tetrahydropyran-2-one |
|
C(C)C1OC(=O)CC(C1)O | 0.0007 |
| FDBF00461 | [(1S,3R,7R,8aR)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] formate |

|
O(C=O)C1C2CC(C=CC2=CC(C1)C)C | 0.0007 |
| FDBF00462 | (4R)-4-hydroxytetrahydropyran-2-one |
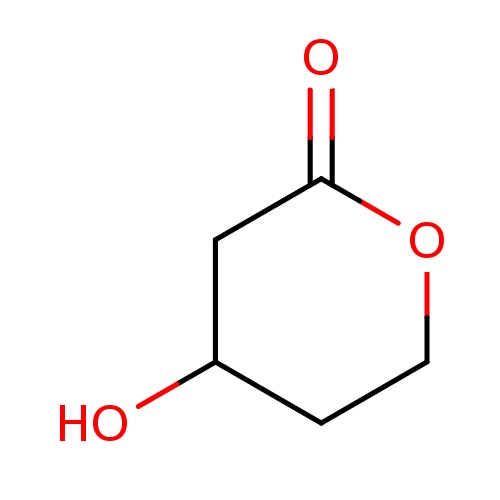
|
O1CCC(CC1=O)O | 0.0007 |
| FDBF01465 | [(1S,3R,7R,8aR)-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] 2-methylpropanoate |
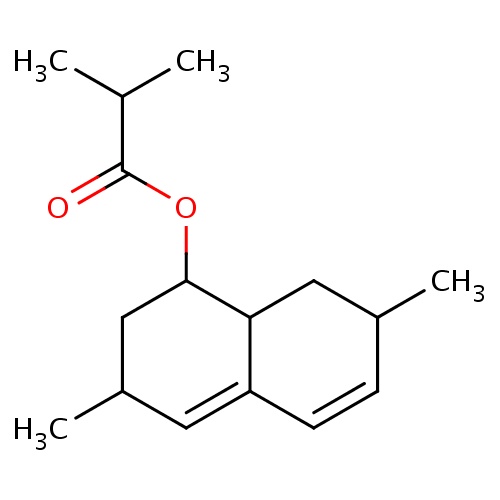
|
O(C(=O)C(C)C)C1C2CC(C=CC2=CC(C1)C)C | 0.0003 |
| FDBF01466 | [(1S,3R,7S,8S,8aR)-3,7,8-trimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] 2-methylpropanoate |
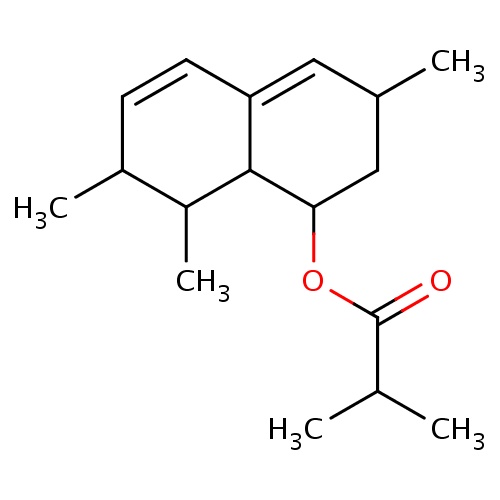
|
CC1C2C(=CC(CC2OC(=O)C(C)C)C)C=CC1C | 0.0003 |

