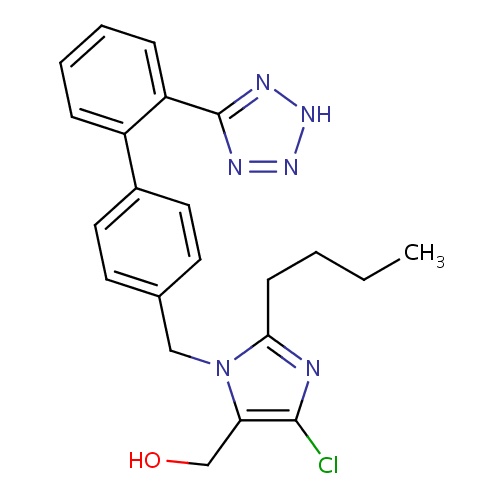
IUPAC name
[2-butyl-4-chloro-1-({4-[2-(2H-1,2,3,4-tetrazol-5-yl)phenyl]phenyl}methyl)-1H-imidazol-5-yl]methanol
SMILES
CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=C1)C1=CC=CC=C1C1=NNN=N1
Compound class
Antihypertensive Agents; Anti-Arrhythmia Agents; Angiotensin II Type 1 Receptor Blockers; Angiotensin Receptor Antagonists; Cardiovascular System; Angiotensin II Antagonists, Plain; Agents Acting on the Renin-Angiotensin System; Angiotensin II Antagonists and Diuretics; Angiotensin II Antagonists and Calcium Channel Blockers; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Angiotensin II Receptor Antagonists; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
May be used as a first line agent to treat uncomplicated hypertension, isolated systolic hypertension and left ventricular hypertrophy. May be used as a first line agent to delay progression of diabetic nephropathy. Losartan may be also used as a second line agent in the treatment of congestive heart failure, systolic dysfunction, myocardial infarction and coronary artery disease in those intolerant of ACE inhibitors.
Common name
Losartan
IUPAC name
[2-butyl-4-chloro-1-({4-[2-(2H-1,2,3,4-tetrazol-5-yl)phenyl]phenyl}methyl)-1H-imidazol-5-yl]methanol
SMILES
CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=C1)C1=CC=CC=C1C1=NNN=N1
INCHI
InChI=1S/C22H23ClN6O/c1-2-3-8-20-24-21(23)19(14-30)29(20)13-15-9-11-16(12-10-15)17-6-4-5-7-18(17)22-25-27-28-26-22/h4-7,9-12,30H,2-3,8,13-14H2,1H3,(H,25,26,27,28)
FORMULA
C22H23ClN6O

Common name
Losartan
IUPAC name
[2-butyl-4-chloro-1-({4-[2-(2H-1,2,3,4-tetrazol-5-yl)phenyl]phenyl}methyl)-1H-imidazol-5-yl]methanol
Molecular weight
422.911
clogP
4.942
clogS
-7.197
HBond Acceptor
5
HBond Donor
2
Total Polar Surface Area
92.51
Number of Rings
4
Rotatable Bond
8
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
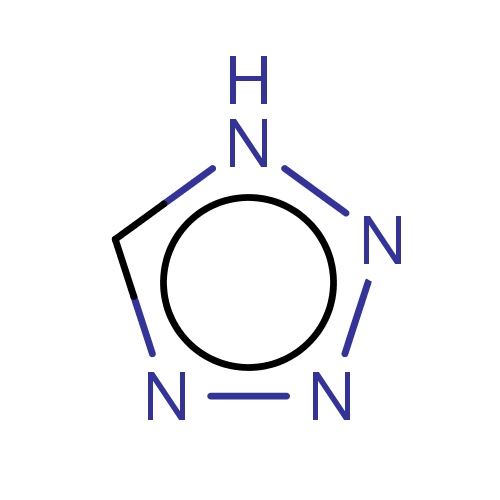
| FDBF00188 | tetrazole |
|
[nH]1nnnc1 | 0.0055 |
| FDBF01565 | (5-chloro-1H-imidazol-4-yl)methanol |

|
Clc1[nH]cnc1CO | 0.0003 |
| FDBF01566 | 4-chloro-1,2-dimethyl-imidazole |

|
Cn1c(nc(c1)Cl)C | 0.0003 |
| FDBF01567 | (5-chloro-2-methyl-1H-imidazol-4-yl)methanol |

|
Cc1[nH]c(c(n1)CO)Cl | 0.0003 |
| FDBF01568 | 1-benzyl-4-chloro-imidazole |

|
C(n1cnc(c1)Cl)c2ccccc2 | 0.0003 |
| FDBF01570 | 5-chloro-2-ethyl-1H-imidazole |

|
C(c1[nH]c(cn1)Cl)C | 0.0003 |
| FDBF01576 | (5-chloro-2-ethyl-1H-imidazol-4-yl)methanol |

|
C(c1[nH]c(c(n1)CO)Cl)C | 0.0003 |
| FDBF01578 | 1-benzyl-4-chloro-2-ethyl-imidazole |

|
C(n1c(nc(c1)Cl)CC)c2ccccc2 | 0.0003 |
| FDBF01580 | (5-chloro-2-ethyl-3-methyl-imidazol-4-yl)methanol |

|
Cn1c(nc(c1CO)Cl)CC | 0.0003 |

