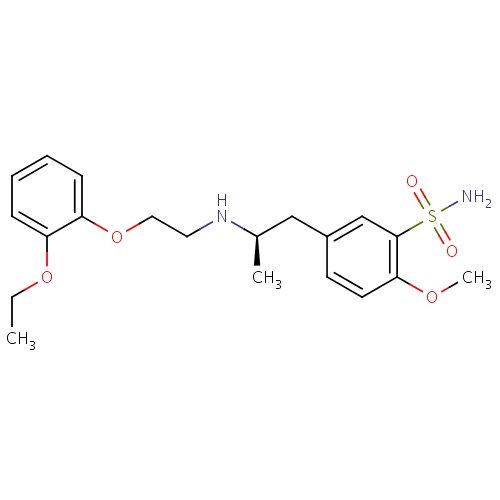
IUPAC name
5-[(2R)-2-{[2-(2-ethoxyphenoxy)ethyl]amino}propyl]-2-methoxybenzene-1-sulfonamide
SMILES
CCOC1=CC=CC=C1OCCN[C@H](C)CC1=CC(=C(OC)C=C1)S(N)(=O)=O
Compound class
Adrenergic alpha-1 Receptor Antagonists; Adrenergic alpha-Antagonists; Genito Urinary System and Sex Hormones; Drugs Used in Benign Prostatic Hypertrophy; Urological Agents; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
Used in the treatment of signs and symptoms of benign prostatic hyperplasia (reduction in urinary obstruction and relief of associated manifestations such as hesitancy, terminal dribbling of urine, interrupted or weak stream..etc.).
Common name
Tamsulosin
IUPAC name
5-[(2R)-2-{[2-(2-ethoxyphenoxy)ethyl]amino}propyl]-2-methoxybenzene-1-sulfonamide
SMILES
CCOC1=CC=CC=C1OCCN[C@H](C)CC1=CC(=C(OC)C=C1)S(N)(=O)=O
INCHI
InChI=1S/C20H28N2O5S/c1-4-26-17-7-5-6-8-18(17)27-12-11-22-15(2)13-16-9-10-19(25-3)20(14-16)28(21,23)24/h5-10,14-15,22H,4,11-13H2,1-3H3,(H2,21,23,24)/t15-/m1/s1
FORMULA
C20H28N2O5S

Common name
Tamsulosin
IUPAC name
5-[(2R)-2-{[2-(2-ethoxyphenoxy)ethyl]amino}propyl]-2-methoxybenzene-1-sulfonamide
Molecular weight
408.512
clogP
2.601
clogS
-5.438
HBond Acceptor
5
HBond Donor
3
Total Polar Surface Area
99.88
Number of Rings
2
Rotatable Bond
11
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00040 | ethanamine |
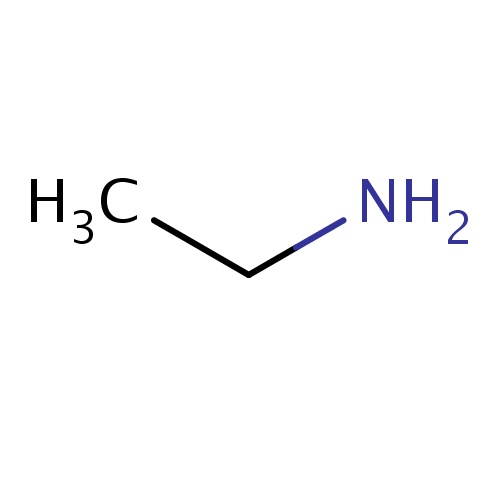
|
CCN | 0.0677 |
| FDBF00041 | ethanol |
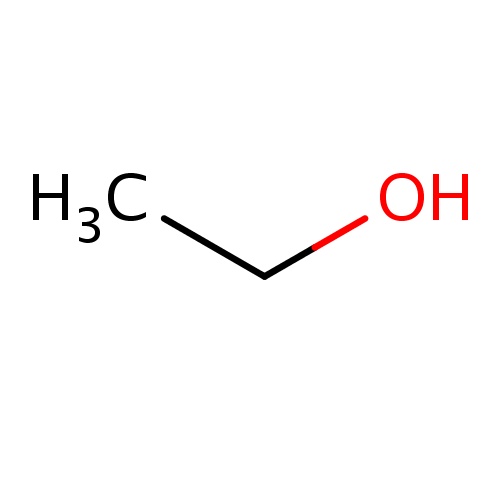
|
CCO | 0.1474 |
| FDBF00203 | benzene-1,2-diol |
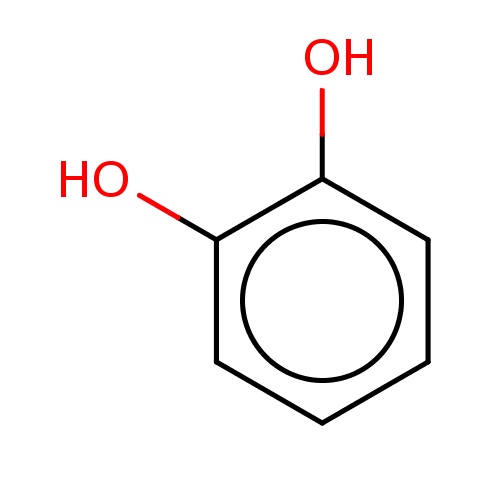
|
c1cc(c(cc1)O)O | 0.0079 |
| FDBF00217 | 2-(isopropylamino)ethanol |
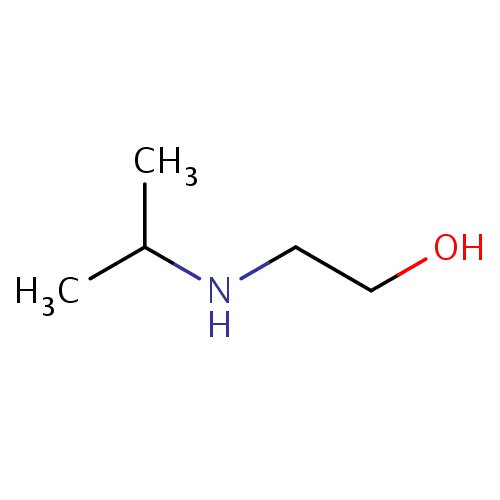
|
C(NC(C)C)CO | 0.0086 |
| FDBF00218 | 1-methoxy-4-methyl-benzene |
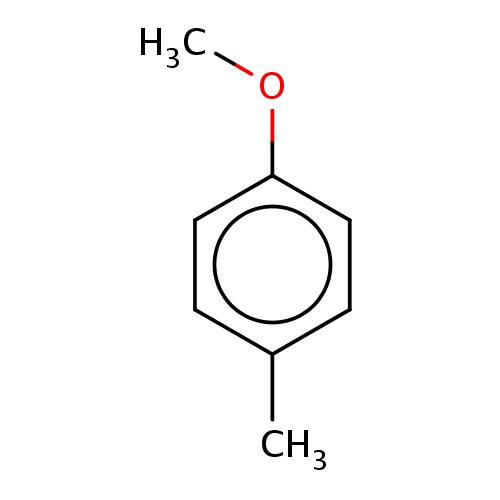
|
O(C)c1ccc(cc1)C | 0.0113 |
| FDBF00243 | N-ethylpropan-2-amine |
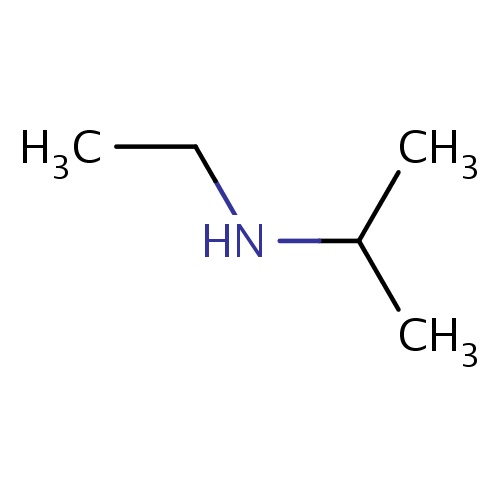
|
C(C)(C)NCC | 0.0045 |
| FDBF01638 | 1-methoxy-4-propyl-benzene |

|
O(C)c1ccc(cc1)CCC | 0.0010 |
| FDBF01640 | 2-phenoxyethanamine |

|
C(CN)Oc1ccccc1 | 0.0014 |
| FDBF01641 | (2R)-1-(4-methoxyphenyl)propan-2-amine |

|
O(C)c1ccc(cc1)CC(C)N | 0.0010 |

