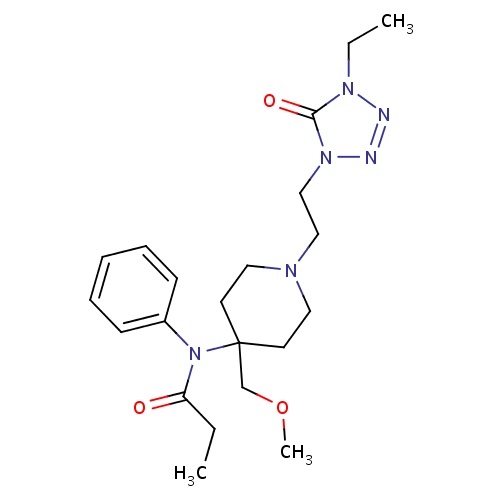
IUPAC name
N-{1-[2-(4-ethyl-5-oxo-4,5-dihydro-1H-1,2,3,4-tetrazol-1-yl)ethyl]-4-(methoxymethyl)piperidin-4-yl}-N-phenylpropanamide
SMILES
CCN1N=NN(CCN2CCC(COC)(CC2)N(C(=O)CC)C2=CC=CC=C2)C1=O
Compound class
Anesthetics, Intravenous; Analgesics, Opioid; Narcotics; Anesthetics; Nervous System; Anesthetics, General; Opioid Anesthetics; CYP3A4 Inhibitors;
Therapeutic area
For the management of postoperative pain and the maintenance of general anesthesia.
Common name
Alfentanil
IUPAC name
N-{1-[2-(4-ethyl-5-oxo-4,5-dihydro-1H-1,2,3,4-tetrazol-1-yl)ethyl]-4-(methoxymethyl)piperidin-4-yl}-N-phenylpropanamide
SMILES
CCN1N=NN(CCN2CCC(COC)(CC2)N(C(=O)CC)C2=CC=CC=C2)C1=O
INCHI
InChI=1S/C21H32N6O3/c1-4-19(28)27(18-9-7-6-8-10-18)21(17-30-3)11-13-24(14-12-21)15-16-26-20(29)25(5-2)22-23-26/h6-10H,4-5,11-17H2,1-3H3
FORMULA
C21H32N6O3

Common name
Alfentanil
IUPAC name
N-{1-[2-(4-ethyl-5-oxo-4,5-dihydro-1H-1,2,3,4-tetrazol-1-yl)ethyl]-4-(methoxymethyl)piperidin-4-yl}-N-phenylpropanamide
Molecular weight
416.517
clogP
1.083
clogS
-3.339
HBond Acceptor
6
HBond Donor
0
Total Polar Surface Area
85.49
Number of Rings
3
Rotatable Bond
10
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00003 | formamide |
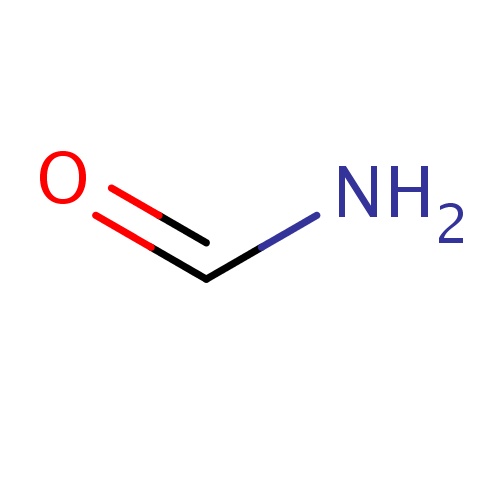
|
C(=O)N | 0.1240 |
| FDBF00671 | 1-ethylpiperidine |

|
N1(CCCCC1)CC | 0.0106 |
| FDBF01366 | N-(1-ethyl-4-piperidyl)formamide |

|
CCN1CCC(CC1)NC=O | 0.0024 |
| FDBF01645 | N-phenyl-N-(4-piperidyl)formamide |

|
c1(ccccc1)N(C2CCNCC2)C=O | 0.0014 |
| FDBF01647 | N-(4-piperidyl)propanamide |

|
N1CCC(CC1)NC(=O)CC | 0.0014 |
| FDBF01651 | N-(1-methyl-4-piperidyl)-N-phenyl-formamide |

|
c1(ccccc1)N(C2CCN(CC2)C)C=O | 0.0014 |
| FDBF01654 | 4-(methoxymethyl)-1-methyl-piperidine |

|
CN1CCC(CC1)COC | 0.0007 |
| FDBF01655 | 1-ethyl-4-methyl-piperidine |

|
CCN1CCC(CC1)C | 0.0017 |
| FDBF01659 | N-[4-(methoxymethyl)-1-methyl-4-piperidyl]formamide |

|
CN1CCC(CC1)(COC)NC=O | 0.0007 |
| FDBF01662 | N-(1,4-dimethyl-4-piperidyl)propanamide |

|
C(C)C(=O)NC1(CCN(CC1)C)C | 0.0007 |

