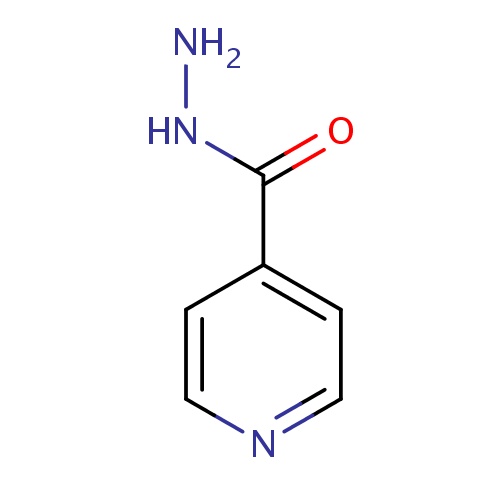
IUPAC name
pyridine-4-carbohydrazide
SMILES
NNC(=O)C1=CC=NC=C1
Compound class
Antitubercular Agents; Fatty Acid Synthesis Inhibitors; Antimycobacterials; Antiinfectives for Systemic Use; Drugs for Treatment of Tuberculosis; Hydrazides; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
For the treatment of all forms of tuberculosis in which organisms are susceptible.
Common name
Isoniazid
IUPAC name
pyridine-4-carbohydrazide
SMILES
NNC(=O)C1=CC=NC=C1
INCHI
InChI=1S/C6H7N3O/c7-9-6(10)5-1-3-8-4-2-5/h1-4H,7H2,(H,9,10)
FORMULA
C6H7N3O

Common name
Isoniazid
IUPAC name
pyridine-4-carbohydrazide
Molecular weight
137.139
clogP
-0.273
clogS
-1.031
HBond Acceptor
2
HBond Donor
3
Total Polar Surface Area
68.01
Number of Rings
1
Rotatable Bond
1