
IUPAC name
[(2R,3R,4S,5R,6S)-3,4,5-tris(acetyloxy)-6-{[(triethyl-λ
SMILES
CCP(CC)(CC)=[Au]S[C@@H]1O[C@H](COC(C)=O)[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O
Compound class
Antirheumatic Agents; Musculo-Skeletal System; Antiinflammatory and Antirheumatic Products; Specific Antirheumatic Agents; Gold Preparations;
Therapeutic area
Used in the treatment of active, progressive or destructive forms of inflammatory arthritis, such as adult rheumatoid arthritis.
Common name
Auranofin
IUPAC name
[(2R,3R,4S,5R,6S)-3,4,5-tris(acetyloxy)-6-{[(triethyl-λ
SMILES
CCP(CC)(CC)=[Au]S[C@@H]1O[C@H](COC(C)=O)[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O
INCHI
InChI=1S/C14H20O9S.C6H15P.Au/c1-6(15)19-5-10-11(20-7(2)16)12(21-8(3)17)13(14(24)23-10)22-9(4)18;1-4-7(5-2)6-3;/h10-14,24H,5H2,1-4H3;4-6H2,1-3H3;/q;;+1/p-1/t10-,11-,12+,13-,14+;;/m1../s1
FORMULA
C20H34AuO9PS

Common name
Auranofin
IUPAC name
[(2R,3R,4S,5R,6S)-3,4,5-tris(acetyloxy)-6-{[(triethyl-λ
Molecular weight
678.484
clogP
2.604
clogS
-3.435
HBond Acceptor
9
HBond Donor
0
Total Polar Surface Area
149.54
Number of Rings
1
Rotatable Bond
14
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
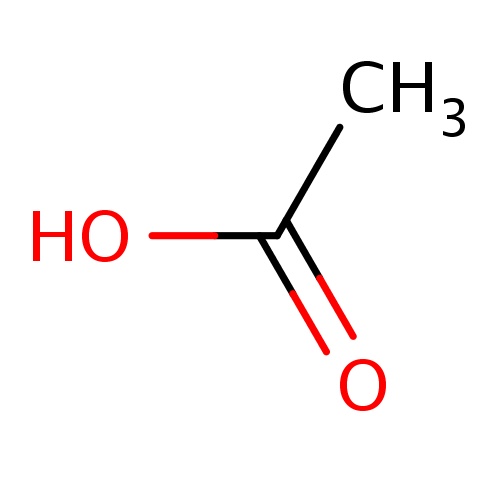
| FDBF00004 | acetic acid |
|
CC(=O)O | 0.0687 |
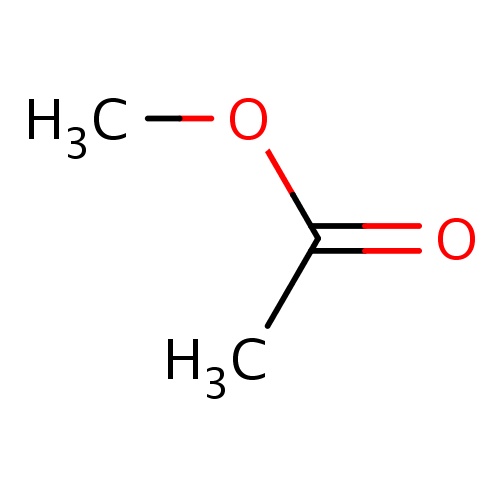
| FDBF00101 | methyl acetate |
|
O(C(=O)C)C | 0.0151 |
| FDBF01354 | (2R)-2-methyltetrahydropyran |

|
C1C(OCCC1)C | 0.0017 |
| FDBF02239 | tetrahydropyran-4-yl acetate |

|
O(C(=O)C)C1CCOCC1 | 0.0003 |
| FDBF02240 | [(3R)-tetrahydropyran-3-yl] acetate |

|
O(C(=O)C)C1CCCOC1 | 0.0003 |
| FDBF02242 | (2S)-tetrahydropyran-2-thiol |

|
SC1CCCCO1 | 0.0003 |
| FDBF02243 | [(3R,6R)-6-methyltetrahydropyran-3-yl] acetate |

|
CC1OCC(CC1)OC(=O)C | 0.0007 |
| FDBF02244 | [(2R,3S)-2-methyltetrahydropyran-3-yl] acetate |

|
CC1OCCCC1OC(=O)C | 0.0003 |
| FDBF02245 | (2S,6R)-6-methyltetrahydropyran-2-thiol |

|
CC1OC(CCC1)S | 0.0003 |
| FDBF02247 | [(2R,4R)-2-methyltetrahydropyran-4-yl] acetate |

|
O(C(=O)C)C1CCOC(C1)C | 0.0003 |

