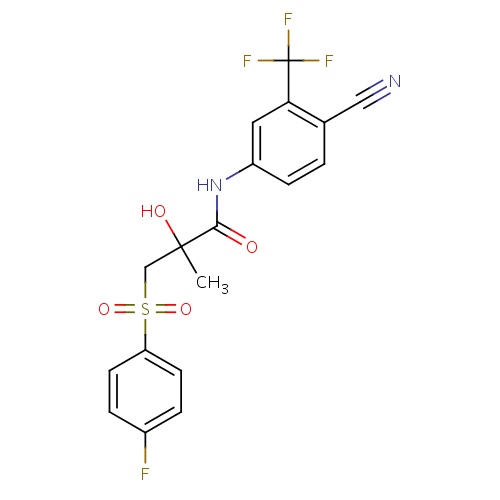
IUPAC name
N-[4-cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorobenzenesulfonyl)-2-hydroxy-2-methylpropanamide
SMILES
CC(O)(CS(=O)(=O)C1=CC=C(F)C=C1)C(=O)NC1=CC(=C(C=C1)C#N)C(F)(F)F
Compound class
Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Anti-Androgens; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
For treatment (together with surgery or LHRH analogue) of advanced prostatic cancer.
Common name
Bicalutamide
IUPAC name
N-[4-cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorobenzenesulfonyl)-2-hydroxy-2-methylpropanamide
SMILES
CC(O)(CS(=O)(=O)C1=CC=C(F)C=C1)C(=O)NC1=CC(=C(C=C1)C#N)C(F)(F)F
INCHI
InChI=1S/C18H14F4N2O4S/c1-17(26,10-29(27,28)14-6-3-12(19)4-7-14)16(25)24-13-5-2-11(9-23)15(8-13)18(20,21)22/h2-8,26H,10H2,1H3,(H,24,25)
FORMULA
C18H14F4N2O4S

Common name
Bicalutamide
IUPAC name
N-[4-cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorobenzenesulfonyl)-2-hydroxy-2-methylpropanamide
Molecular weight
430.373
clogP
3.224
clogS
-5.193
HBond Acceptor
5
HBond Donor
2
Total Polar Surface Area
107.26
Number of Rings
2
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00003 | formamide |
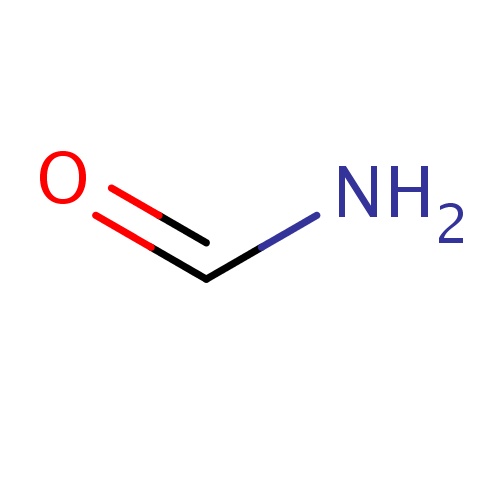
|
C(=O)N | 0.1240 |
| FDBF00042 | propan-2-ol |
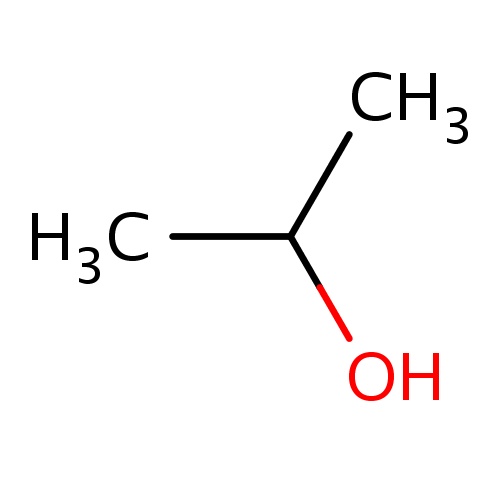
|
CC(O)C | 0.0278 |
| FDBF00177 | fluoroform |
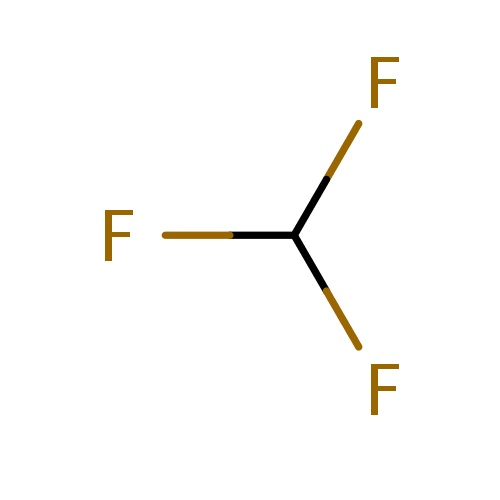
|
FC(F)F | 0.0704 |
| FDBF00283 | BLAH |
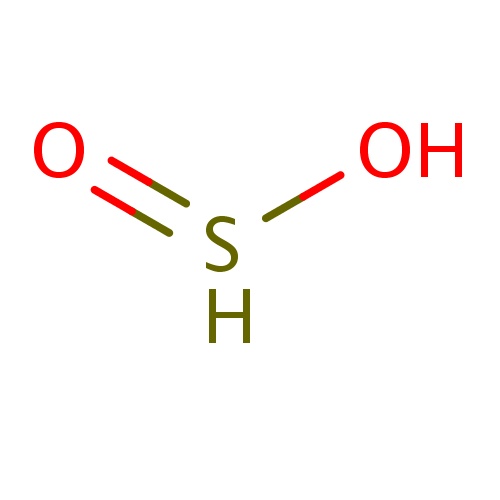
|
S(=O)O | 0.0244 |
| FDBF00378 | fluorobenzene |
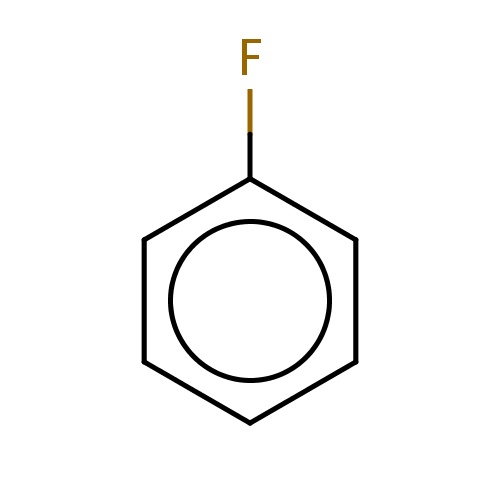
|
Fc1ccccc1 | 0.0237 |
| FDBF02254 | benzonitrile |
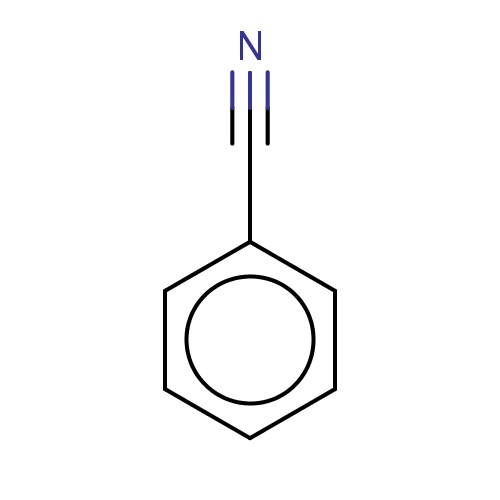
|
c1ccc(cc1)C#N | 0.0041 |
| FDBF02550 | 1-(dihydroxy-λ3-sulfanyl)-4-fluoro-benzene |
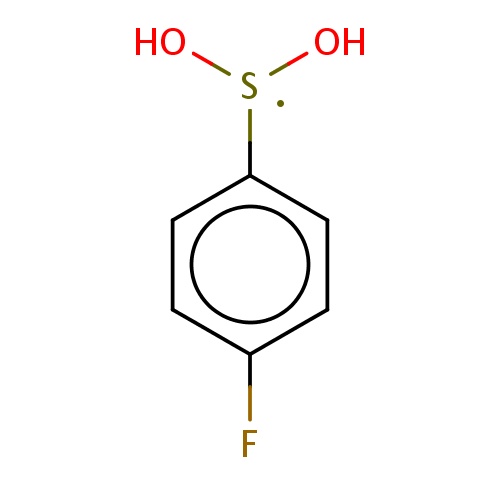
|
[S](O)(O)c1ccc(cc1)F | 0.0003 |
| FDBF02551 | N-(4-cyanophenyl)formamide |
|
c1(ccc(cc1)C#N)NC=O | 0.0003 |
| FDBF02552 | 2-(trifluoromethyl)benzonitrile |
|
c1cc(c(cc1)C#N)C(F)(F)F | 0.0007 |
| FDBF02554 | N-[4-cyano-3-(trifluoromethyl)phenyl]formamide |
|
c1(cc(c(cc1)C#N)C(F)(F)F)NC=O | 0.0003 |

