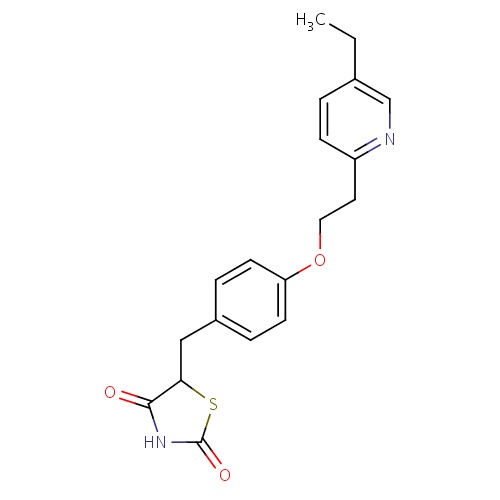
IUPAC name
5-({4-[2-(5-ethylpyridin-2-yl)ethoxy]phenyl}methyl)-1,3-thiazolidine-2,4-dione
SMILES
CCC1=CN=C(CCOC2=CC=C(CC3SC(=O)NC3=O)C=C2)C=C1
Compound class
Hypoglycemic Agents; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Thiazolidinediones; Blood Glucose Lowering Drugs, Excl. Insulins; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
Treatment of Type II diabetes mellitus.
Common name
Pioglitazone
IUPAC name
5-({4-[2-(5-ethylpyridin-2-yl)ethoxy]phenyl}methyl)-1,3-thiazolidine-2,4-dione
SMILES
CCC1=CN=C(CCOC2=CC=C(CC3SC(=O)NC3=O)C=C2)C=C1
INCHI
InChI=1S/C19H20N2O3S/c1-2-13-3-6-15(20-12-13)9-10-24-16-7-4-14(5-8-16)11-17-18(22)21-19(23)25-17/h3-8,12,17H,2,9-11H2,1H3,(H,21,22,23)
FORMULA
C19H20N2O3S

Common name
Pioglitazone
IUPAC name
5-({4-[2-(5-ethylpyridin-2-yl)ethoxy]phenyl}methyl)-1,3-thiazolidine-2,4-dione
Molecular weight
356.439
clogP
4.279
clogS
-5.725
HBond Acceptor
4
HBond Donor
1
Total Polar Surface Area
93.59
Number of Rings
3
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
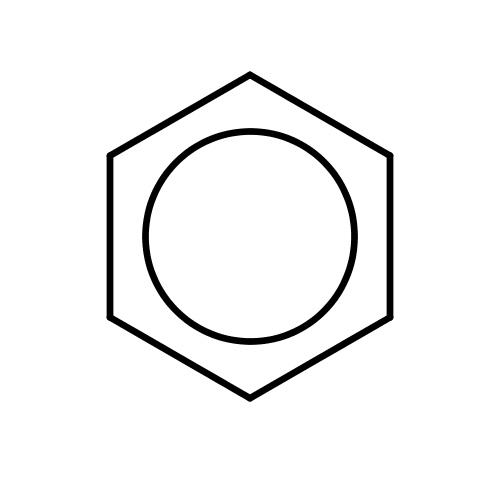
| FDBF00005 | benzene |
|
c1ccccc1 | 0.2824 |
| FDBF00014 | pyridine |
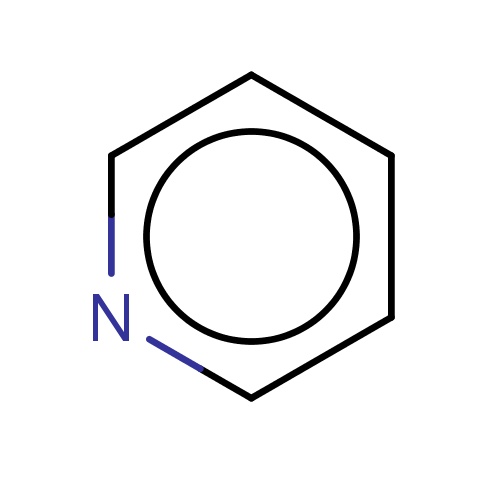
|
c1cccnc1 | 0.0333 |
| FDBF00041 | ethanol |
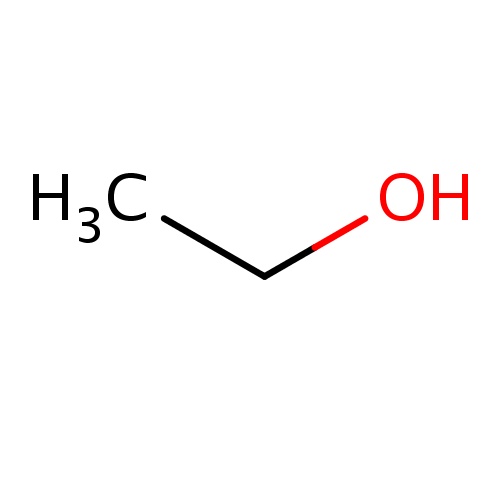
|
CCO | 0.1474 |
| FDBF00088 | p-cresol |
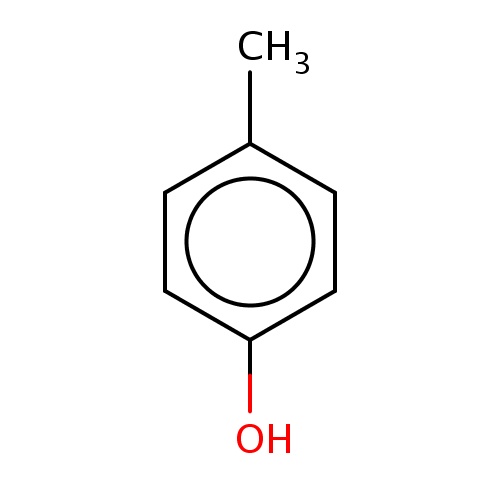
|
Cc1ccc(cc1)O | 0.0134 |
| FDBF00218 | 1-methoxy-4-methyl-benzene |
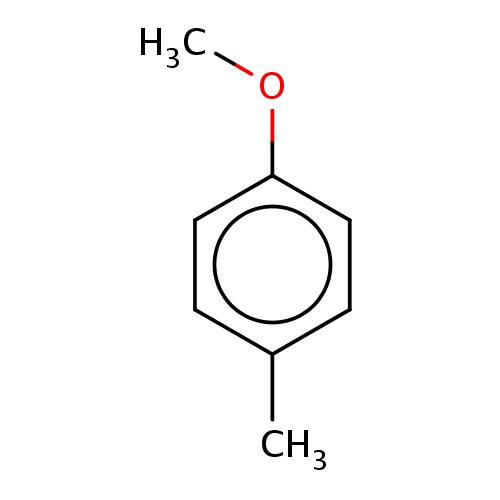
|
O(C)c1ccc(cc1)C | 0.0113 |
| FDBF00361 | 2-methylpyridine |
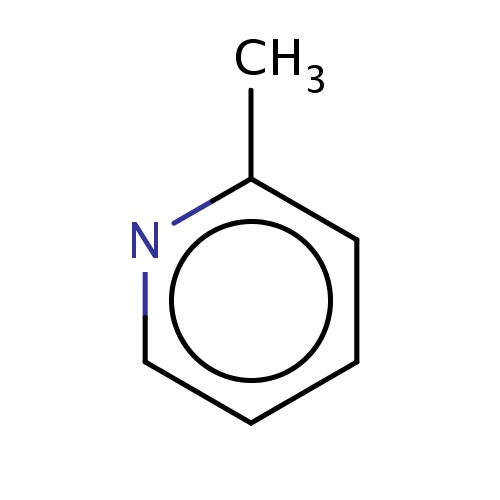
|
n1c(cccc1)C | 0.0058 |
| FDBF00585 | 2-ethylpyridine |
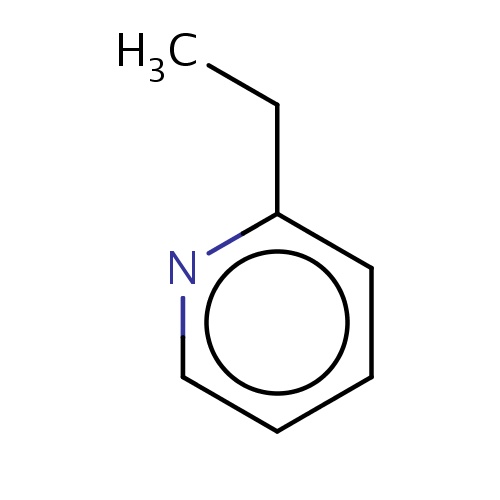
|
n1c(cccc1)CC | 0.0034 |
| FDBF00911 | 1-ethoxy-4-methyl-benzene |

|
O(c1ccc(cc1)C)CC | 0.0031 |
| FDBF02563 | 5-ethyl-2-methyl-pyridine |
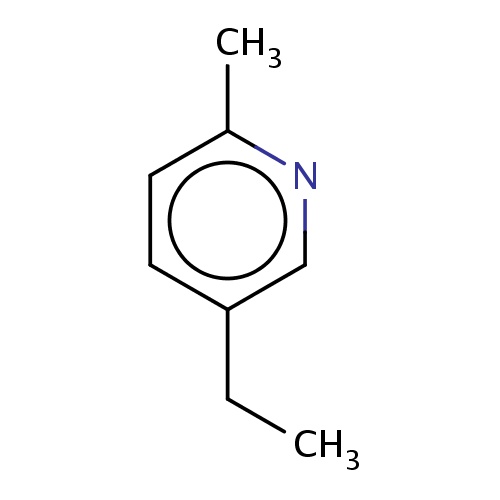
|
Cc1ncc(cc1)CC | 0.0003 |
| FDBF02564 | 2-(2-pyridyl)ethanol |
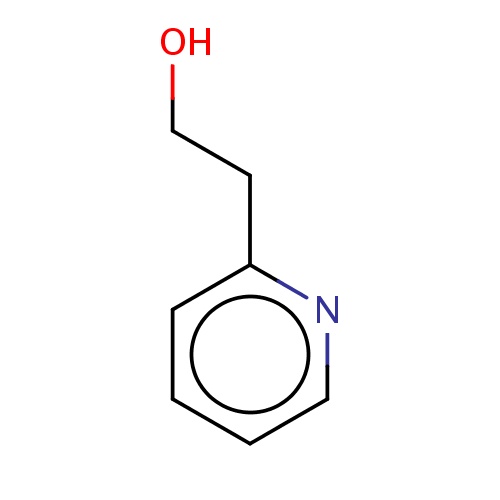
|
C(CO)c1ncccc1 | 0.0003 |

