IUPAC name
(1S,2R,7R,10S,11S,14S,15S)-N-tert-butyl-2,15-dimethyl-5-oxo-6-azatetracyclo[8.7.0.0²,
SMILES
[H][C@@]12CC[C@H](C(=O)NC(C)(C)C)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])NC(=O)C=C[C@]12C
Compound class
Adrenergic alpha-Antagonists; 5-alpha Reductase Inhibitors; Skin and Mucous Membrane Agents; Genito Urinary System and Sex Hormones; Dermatologicals; Drugs Used in Benign Prostatic Hypertrophy; Testosterone-5-Alpha Reductase Inhibitors; Urological Agents; CYP3A4 Inhibitors;
Therapeutic area
For the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: Improve symptoms, reduce the risk of acute urinary retention, reduce the risk of the need for surgery including transurethral resection of the prostate. Also used for the stimulation of regrowth of hair in men with mild to moderate androgenetic alopecia (male pattern alopecia, hereditary alopecia, common male baldness).
Common name
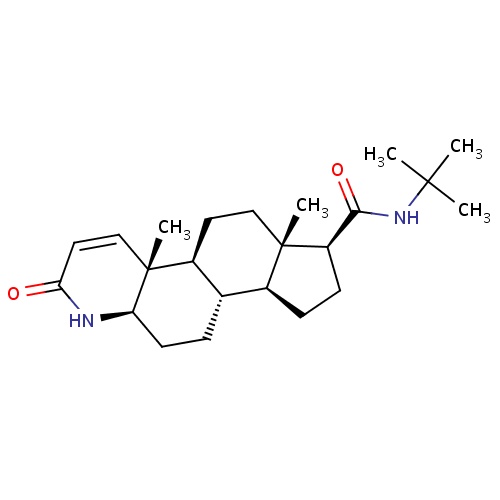
Finasteride
IUPAC name
(1S,2R,7R,10S,11S,14S,15S)-N-tert-butyl-2,15-dimethyl-5-oxo-6-azatetracyclo[8.7.0.0²,
SMILES
[H][C@@]12CC[C@H](C(=O)NC(C)(C)C)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])NC(=O)C=C[C@]12C
INCHI
InChI=1S/C23H36N2O2/c1-21(2,3)25-20(27)17-8-7-15-14-6-9-18-23(5,13-11-19(26)24-18)16(14)10-12-22(15,17)4/h11,13-18H,6-10,12H2,1-5H3,(H,24,26)(H,25,27)/t14-,15-,16-,17+,18+,22-,23+/m0/s1
FORMULA
C23H36N2O2

Common name
Finasteride
IUPAC name
(1S,2R,7R,10S,11S,14S,15S)-N-tert-butyl-2,15-dimethyl-5-oxo-6-azatetracyclo[8.7.0.0²,
Molecular weight
372.544
clogP
3.201
clogS
-4.543
HBond Acceptor
2
HBond Donor
2
Total Polar Surface Area
58.2
Number of Rings
4
Rotatable Bond
2