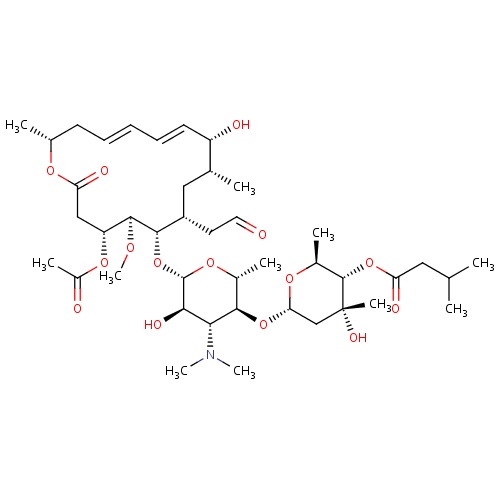
IUPAC name
(2S,3S,4R,6S)-6-{[(2R,3S,4R,5R,6S)-6-{[(4R,5S,6S,7R,9R,10R,11E,13E,16R)-4-(acetyloxy)-10-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-7-(2-oxoethyl)-1-oxacyclohexadeca-11,13-dien-6-yl]oxy}-4-(dimethylamino)-5-hydroxy-2-methyloxan-3-yl]oxy}-4-hydroxy-2,4-dimethyloxan-3-yl 3-methylbutanoate
SMILES
CO[C@H]1[C@H](OC(=O)C)CC(=O)O[C@H](C)C\C=C\C=C\[C@H](O)[C@H](C)C[C@H](CC=O)[C@@H]1O[C@@H]1O[C@H](C)[C@@H](O[C@@H]2O[C@@H](C)[C@H](OC(=O)CC(C)C)[C@](C)(O)C2)[C@H](N(C)C)[C@H]1O
Compound class
Anti-Bacterial Agents; Macrolides; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Macrolides, Lincosamides and Streptogramins; CYP3A4 Inhibitors;
Therapeutic area
For the treatment of bacterial infections.
Common name
Josamycin
IUPAC name
(2S,3S,4R,6S)-6-{[(2R,3S,4R,5R,6S)-6-{[(4R,5S,6S,7R,9R,10R,11E,13E,16R)-4-(acetyloxy)-10-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-7-(2-oxoethyl)-1-oxacyclohexadeca-11,13-dien-6-yl]oxy}-4-(dimethylamino)-5-hydroxy-2-methyloxan-3-yl]oxy}-4-hydroxy-2,4-dimethyloxan-3-yl 3-methylbutanoate
SMILES
CO[C@H]1[C@H](OC(=O)C)CC(=O)O[C@H](C)C\C=C\C=C\[C@H](O)[C@H](C)C[C@H](CC=O)[C@@H]1O[C@@H]1O[C@H](C)[C@@H](O[C@@H]2O[C@@H](C)[C@H](OC(=O)CC(C)C)[C@](C)(O)C2)[C@H](N(C)C)[C@H]1O
INCHI
InChI=1S/C42H69NO15/c1-23(2)19-32(47)56-40-27(6)53-34(22-42(40,8)50)57-37-26(5)54-41(36(49)35(37)43(9)10)58-38-29(17-18-44)20-24(3)30(46)16-14-12-13-15-25(4)52-33(48)21-31(39(38)51-11)55-28(7)45/h12-14,16,18,23-27,29-31,34-41,46,49-50H,15,17,19-22H2,1-11H3/b13-12+,16-14+/t24-,25-,26-,27+,29+,30+,31-,34+,35-,36-,37-,38+,39+,40+,41+,42-/m1/s1
FORMULA
C42H69NO15

Common name
Josamycin
IUPAC name
(2S,3S,4R,6S)-6-{[(2R,3S,4R,5R,6S)-6-{[(4R,5S,6S,7R,9R,10R,11E,13E,16R)-4-(acetyloxy)-10-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-7-(2-oxoethyl)-1-oxacyclohexadeca-11,13-dien-6-yl]oxy}-4-(dimethylamino)-5-hydroxy-2-methyloxan-3-yl]oxy}-4-hydroxy-2,4-dimethyloxan-3-yl 3-methylbutanoate
Molecular weight
827.995
clogP
1.232
clogS
-2.172
HBond Acceptor
16
HBond Donor
3
Total Polar Surface Area
206.05
Number of Rings
3
Rotatable Bond
14
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
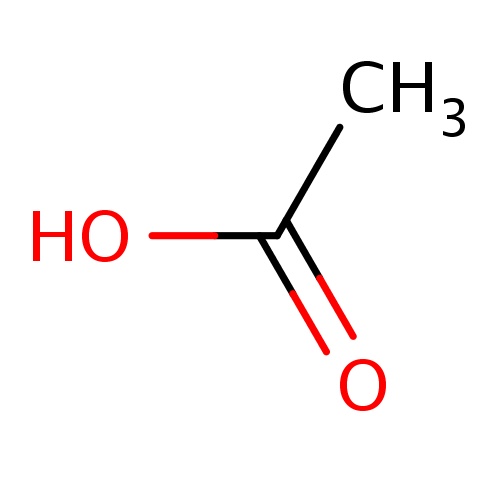
| FDBF00004 | acetic acid |
|
CC(=O)O | 0.0687 |
| FDBF00007 | propane |

|
C(C)C | 0.2412 |
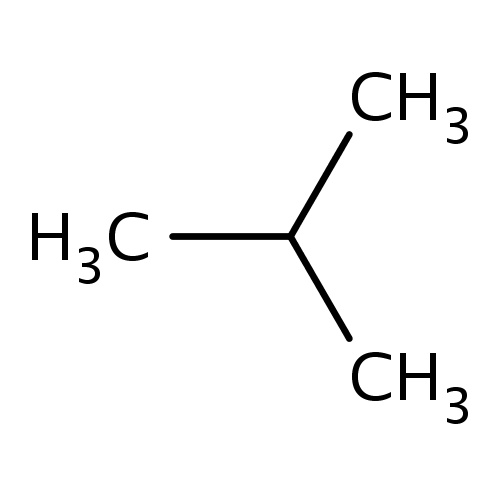
| FDBF00012 | isobutane |
|
C(C)(C)C | 0.0611 |
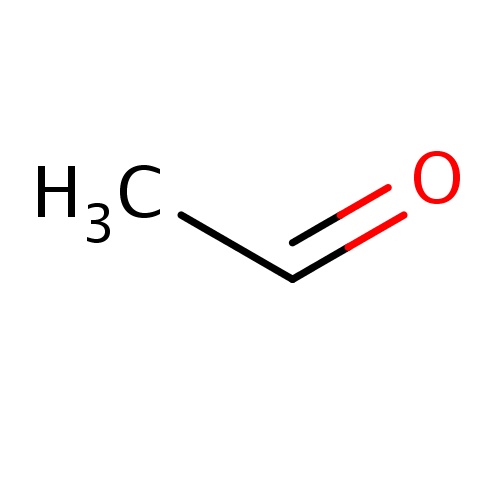
| FDBF00098 | acetaldehyde |
|
CC=O | 0.0182 |
| FDBF02213 | (2S,3S,6S)-6-methyltetrahydropyran-2,3-diol |
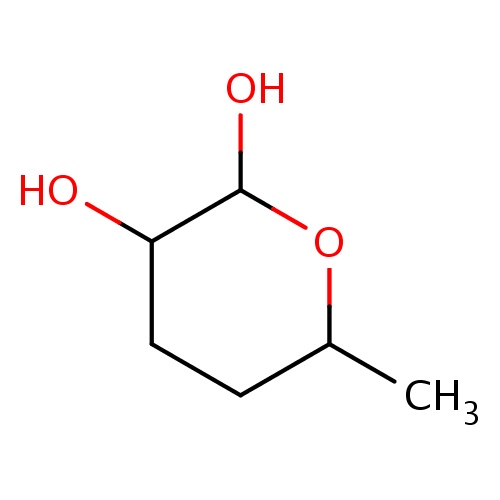
|
OC1OC(CCC1O)C | 0.0007 |
| FDBF02214 | (3S,4R,6S)-4-(dimethylamino)-6-methyl-tetrahydropyran-3-ol |
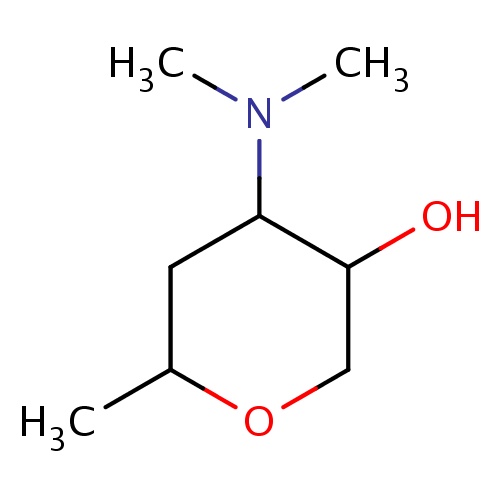
|
N(C)(C)C1C(COC(C1)C)O | 0.0007 |
| FDBF02218 | (3S,6S)-6-methyltetrahydropyran-3-ol |
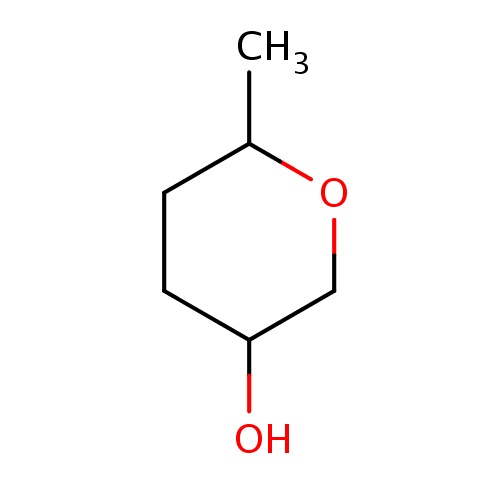
|
O1CC(CCC1C)O | 0.0007 |
| FDBF02934 | (2S,3R,5S)-2-methyltetrahydropyran-3,5-diol |
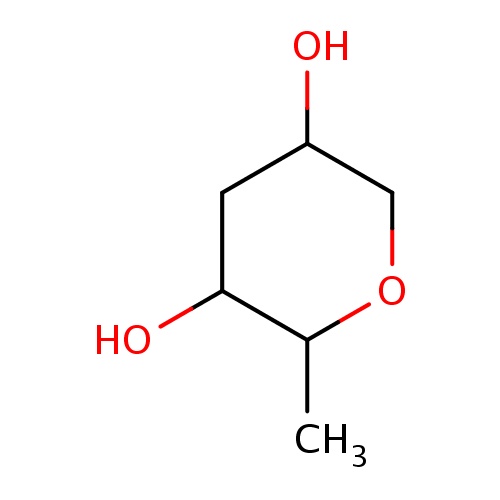
|
O1C(C(CC(C1)O)O)C | 0.0003 |
| FDBF02935 | (3R,5E,7E,9S,10R,12S)-9-hydroxy-3,10,12-trimethyl-2-oxacyclohexadeca-5,7-dien-1-one |

|
CC1CC(C(C=CC=CCC(OC(=O)CCCC1)C)O)C | 0.0003 |
| FDBF02936 | (3R,5E,7E,9S,10R)-9-hydroxy-3,10-dimethyl-2-oxacyclohexadeca-5,7-dien-1-one |

|
OC1C(CCCCCCC(=O)OC(CC=CC=C1)C)C | 0.0003 |

