IUPAC name
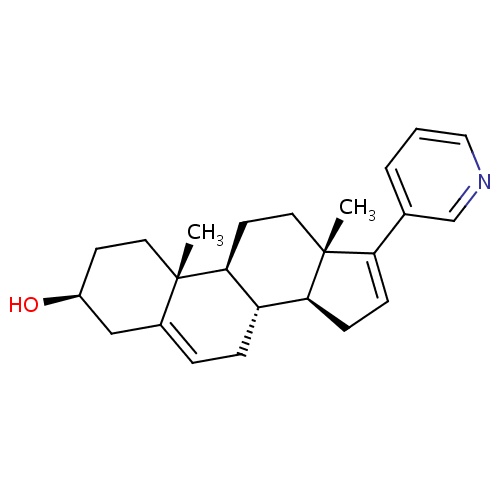
(1S,2R,5S,10R,11S,15S)-2,15-dimethyl-14-(pyridin-3-yl)tetracyclo[8.7.0.0²,
SMILES
[H][C@@]12CC=C(C3=CC=CN=C3)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC=C2C[C@@H](O)CC[C@]12C
Compound class
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; Steroid Synthesis Inhibitors; Cytochrome P-450 Enzyme Inhibitors; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
Used in combination with prednisone for the treatment of metastatic, castration-resistant prostate cancer.
Common name
Abiraterone
IUPAC name
(1S,2R,5S,10R,11S,15S)-2,15-dimethyl-14-(pyridin-3-yl)tetracyclo[8.7.0.0²,
SMILES
[H][C@@]12CC=C(C3=CC=CN=C3)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC=C2C[C@@H](O)CC[C@]12C
INCHI
InChI=1S/C24H31NO/c1-23-11-9-18(26)14-17(23)5-6-19-21-8-7-20(16-4-3-13-25-15-16)24(21,2)12-10-22(19)23/h3-5,7,13,15,18-19,21-22,26H,6,8-12,14H2,1-2H3/t18-,19-,21-,22-,23-,24+/m0/s1
FORMULA
C24H31NO

Common name
Abiraterone
IUPAC name
(1S,2R,5S,10R,11S,15S)-2,15-dimethyl-14-(pyridin-3-yl)tetracyclo[8.7.0.0²,
Molecular weight
349.509
clogP
4.481
clogS
-4.852
HBond Acceptor
2
HBond Donor
1
Total Polar Surface Area
33.12
Number of Rings
5
Rotatable Bond
1