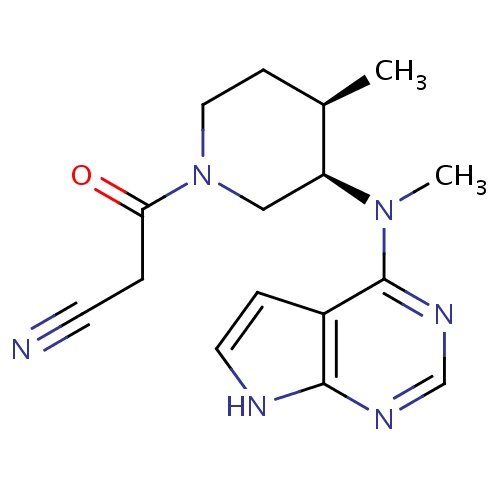
IUPAC name
3-[(3R,4R)-4-methyl-3-[methyl({7H-pyrrolo[2,3-d]pyrimidin-4-yl})amino]piperidin-1-yl]-3-oxopropanenitrile
SMILES
[H][C@@]1(C)CCN(C[C@]1([H])N(C)C1=NC=NC2=C1C=CN2)C(=O)CC#N
Compound class
Therapeutic area
For the treatment of moderate to severe rheumatoid arthritis which is resistant or intolerant to methotrexate therapy. It may also be used as an adjunct to methotrexate therapy, or other non-biologic disease-modifying antirheumatic drugs (DMARDS), when methotrexate alone is not sufficient.
Common name
Tofacitinib
IUPAC name
3-[(3R,4R)-4-methyl-3-[methyl({7H-pyrrolo[2,3-d]pyrimidin-4-yl})amino]piperidin-1-yl]-3-oxopropanenitrile
SMILES
[H][C@@]1(C)CCN(C[C@]1([H])N(C)C1=NC=NC2=C1C=CN2)C(=O)CC#N
INCHI
InChI=1S/C16H20N6O/c1-11-5-8-22(14(23)3-6-17)9-13(11)21(2)16-12-4-7-18-15(12)19-10-20-16/h4,7,10-11,13H,3,5,8-9H2,1-2H3,(H,18,19,20)/t11-,13+/m1/s1
FORMULA
C16H20N6O

Common name
Tofacitinib
IUPAC name
3-[(3R,4R)-4-methyl-3-[methyl({7H-pyrrolo[2,3-d]pyrimidin-4-yl})amino]piperidin-1-yl]-3-oxopropanenitrile
Molecular weight
312.370
clogP
1.041
clogS
-2.882
HBond Acceptor
5
HBond Donor
1
Total Polar Surface Area
88.91
Number of Rings
3
Rotatable Bond
5
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00569 | acetonitrile |
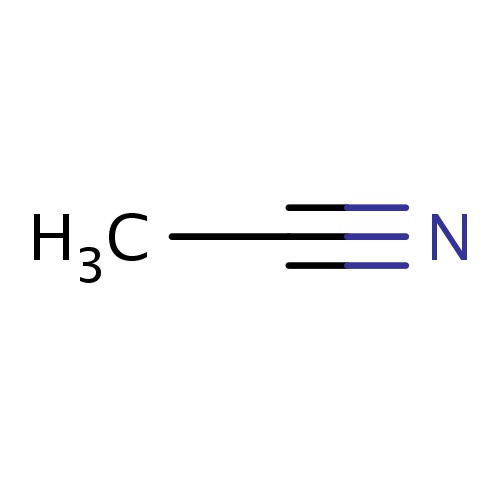
|
CC#N | 0.0161 |
| FDBF03763 | 7H-pyrrolo[2,3-d]pyrimidine |

|
n1cc2c([nH]cc2)nc1 | 0.0007 |
| FDBF03781 | (3R,4R)-4-methyl-3-(methylamino)piperidine-1-carbaldehyde |

|
O=CN1CC(C(CC1)C)NC | 0.0003 |
| FDBF03782 | N-methyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine |

|
N(C)c1ncnc2c1cc[nH]2 | 0.0003 |
| FDBF03783 | 3-(4-methyl-1-piperidyl)-3-oxo-propanenitrile |

|
O=C(N1CCC(CC1)C)CC#N | 0.0003 |
| FDBF03784 | 4-methylpiperidine-1-carbaldehyde |

|
O=CN1CCC(CC1)C | 0.0003 |

