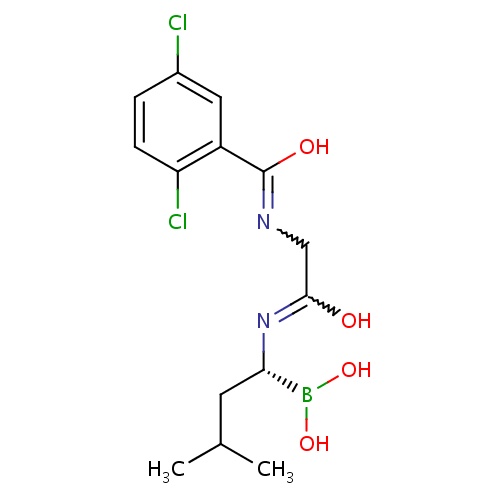
IUPAC name
2-{[(2,5-dichlorophenyl)(hydroxy)methylidene]amino}-N-[(1R)-1-(dihydroxyboranyl)-3-methylbutyl]ethanimidic acid
SMILES
[H][C@@](CC(C)C)(N=C(O)CN=C(O)C1=C(Cl)C=CC(Cl)=C1)B(O)O
Compound class
Antineoplastic Agents; Protease Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
Ixazomib is indicated in combination with lenalidomide and dexamethasone for the treatment of patients with multiple myeloma who have received at least one prior therapy.
Common name
Ixazomib
IUPAC name
2-{[(2,5-dichlorophenyl)(hydroxy)methylidene]amino}-N-[(1R)-1-(dihydroxyboranyl)-3-methylbutyl]ethanimidic acid
SMILES
[H][C@@](CC(C)C)(N=C(O)CN=C(O)C1=C(Cl)C=CC(Cl)=C1)B(O)O
INCHI
InChI=1S/C14H19BCl2N2O4/c1-8(2)5-12(15(22)23)19-13(20)7-18-14(21)10-6-9(16)3-4-11(10)17/h3-4,6,8,12,22-23H,5,7H2,1-2H3,(H,18,21)(H,19,20)/t12-/m0/s1
FORMULA
C14H19BCl2N2O4

Common name
Ixazomib
IUPAC name
2-{[(2,5-dichlorophenyl)(hydroxy)methylidene]amino}-N-[(1R)-1-(dihydroxyboranyl)-3-methylbutyl]ethanimidic acid
Molecular weight
361.029
clogP
1.734
clogS
-2.737
HBond Acceptor
6
HBond Donor
4
Total Polar Surface Area
105.64
Number of Rings
1
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00007 | propane |

|
C(C)C | 0.2412 |
| FDBF00012 | isobutane |
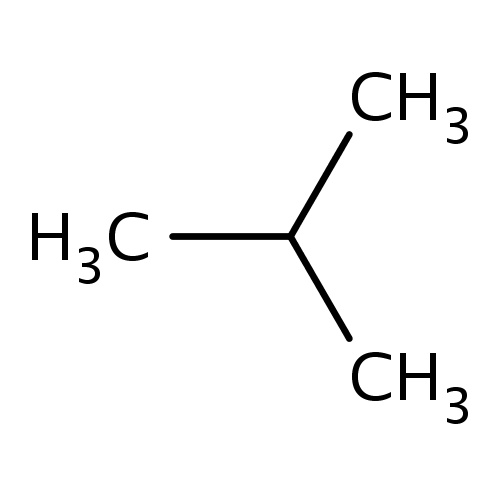
|
C(C)(C)C | 0.0611 |
| FDBF00027 | N-methylacetamide |
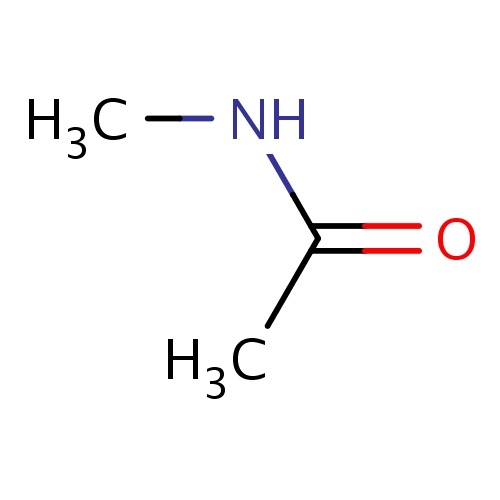
|
N(C(=O)C)C | 0.0175 |
| FDBF00115 | 2-methylbutane |

|
C(C)(C)CC | 0.0103 |
| FDBF00228 | N-ethylacetamide |
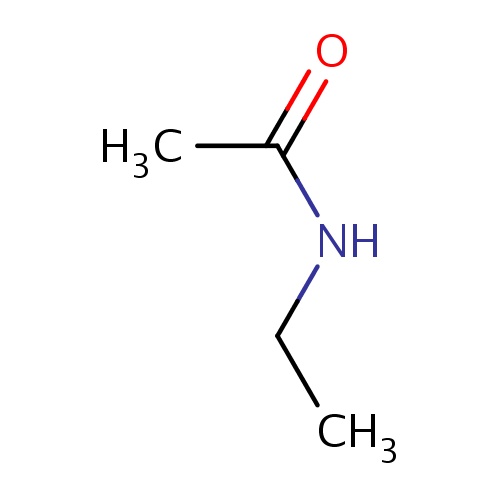
|
C(=O)(NCC)C | 0.0089 |
| FDBF00230 | N-isopentylacetamide |

|
C(C)(C)CCNC(=O)C | 0.0010 |
| FDBF04459 | methylboronic acid |
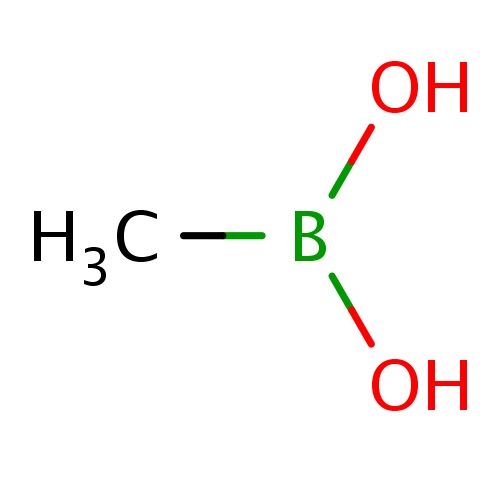
|
B(O)(O)C | 0.0003 |
| FDBF04461 | ethylboronic acid |

|
B(O)(O)CC | 0.0003 |
| FDBF04462 | isopentylboronic acid |

|
B(O)(O)CCC(C)C | 0.0003 |
| FDBF04463 | 2,5-dichlorobenzamide |

|
c1c(cc(c(c1)Cl)C(=O)N)Cl | 0.0003 |

