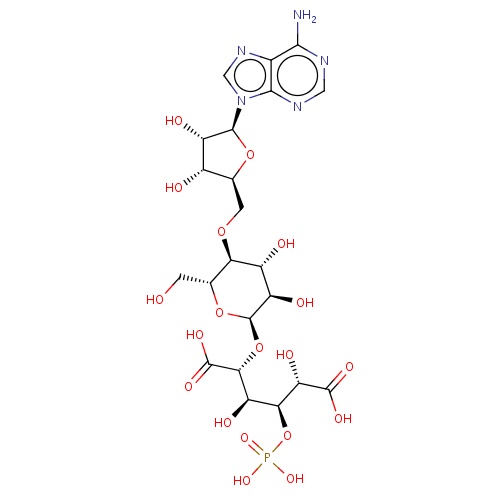
IUPAC name
O-5'-deoxyadenosin-5'-yl-(5'
SMILES
C([C@@H]1[C@H]([C@@H]([C@H]([C@H](O1)O[C@H]([C@@H]([C@@H]([C@@H](C(=O)O)O)OP(=O)(O)O)O)C(=O)O)O)O)OC[C@H]1[C@@H]([C@@H]([C@@H](n2cnc3c(N)ncnc23)O1)O)O)O
Compound class
Insecticide
Therapeutic area
Insecticide
Common name
thuringiensin
IUPAC name
O-5'-deoxyadenosin-5'-yl-(5'
SMILES
C([C@@H]1[C@H]([C@@H]([C@H]([C@H](O1)O[C@H]([C@@H]([C@@H]([C@@H](C(=O)O)O)OP(=O)(O)O)O)C(=O)O)O)O)OC[C@H]1[C@@H]([C@@H]([C@@H](n2cnc3c(N)ncnc23)O1)O)O)O
INCHI
InChI=1S/C22H32N5O19P/c23-17-7-18(25-3-24-17)27(4-26-7)19-10(31)8(29)6(43-19)2-42-14-5(1-28)44-22(11(32)9(14)30)45-16(21(37)38)12(33)15(13(34)20(35)36)46-47(39,40)41/h3-6,8-16,19,22,28-34H,1-2H2,(H,35,36)(H,37,38)(H2,23,24,25)(H2,39,40,41)/t5-,6+,8+,9-,10+,11-,12-,13+,14-,15+,16-,19+,22-/m1/s1
FORMULA
C22H32N5O19P

Common name
thuringiensin
IUPAC name
O-5'-deoxyadenosin-5'-yl-(5'
Molecular weight
701.485
clogP
-7.735
clogS
6.046
HBond Acceptor
22
HBond Donor
13
Total Polar Surface Area
389.51
Number of Rings
4
Rotatable Bond
14
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
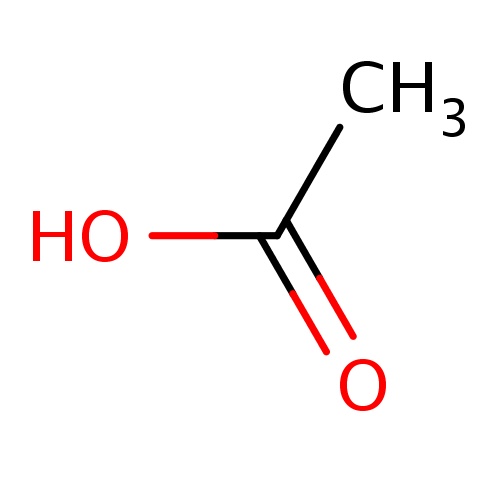
| FDBF00004 | acetic acid |
|
CC(=O)O | 0.0687 |
| FDBF00041 | ethanol |
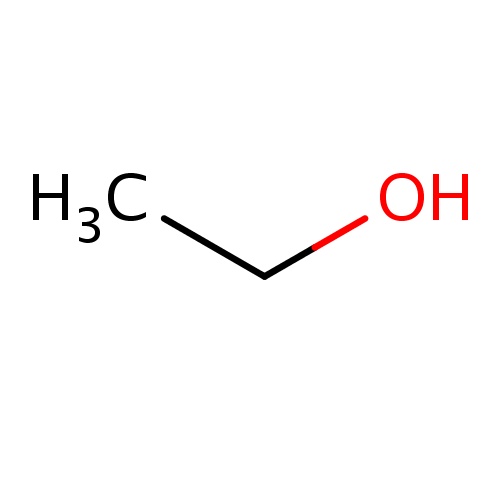
|
CCO | 0.1474 |
| FDBF00059 | (3S,4R)-tetrahydrofuran-3,4-diol |
|
O1CC(C(C1)O)O | 0.0048 |
| FDBF00149 | 3-hydroxypropanoic acid |
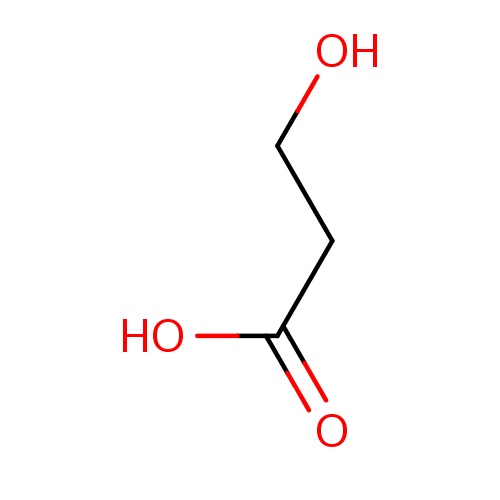
|
C(O)CC(=O)O | 0.0038 |
| FDBF00152 | (3R)-3-hydroxybutanoic acid |
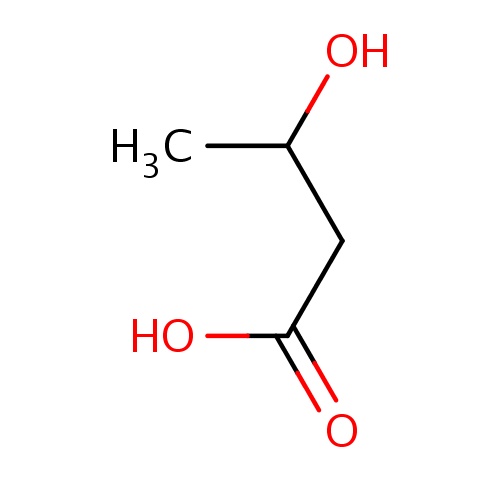
|
C(O)(C)CC(=O)O | 0.0017 |
| FDBF00609 | (3S,4S)-tetrahydropyran-3,4-diol |

|
O1CCC(C(C1)O)O | 0.0021 |
| FDBF00723 | 2-hydroxyacetic acid |
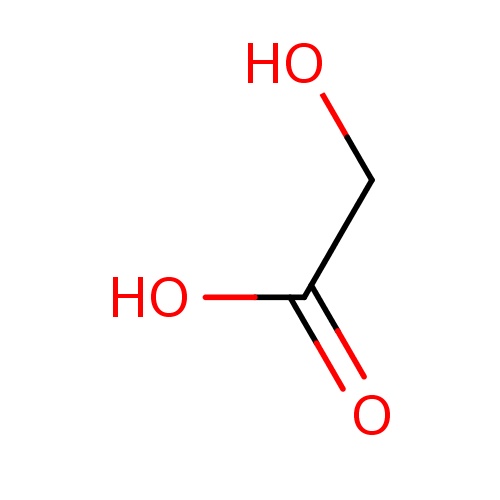
|
OCC(=O)O | 0.0086 |
| FDBF03150 | (2S)-2-hydroxypropanoic acid |
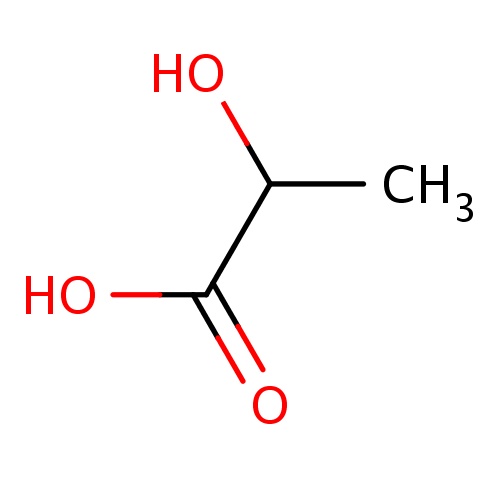
|
C(O)(C)C(=O)O | 0.0069 |
| FDBF05539 | (2S,3R,4R)-2-methyloxolane-3,4-diol |

|
C[C@@H]1OC[C@H]([C@H]1O)O | 0.0003 |

