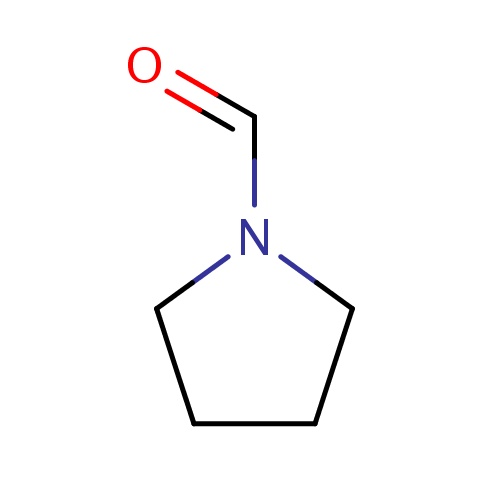
Common name
pyrrolidine-1-carbaldehyde
IUPAC name
pyrrolidine-1-carbaldehyde
SMILES
C(=O)N1CCCC1
Common name
pyrrolidine-1-carbaldehyde
IUPAC name
pyrrolidine-1-carbaldehyde
SMILES
C(=O)N1CCCC1
INCHI
InChI=1S/C5H9NO/c7-5-6-3-1-2-4-6/h5H,1-4H2
FORMULA
C5H9NO

Common name
pyrrolidine-1-carbaldehyde
IUPAC name
pyrrolidine-1-carbaldehyde
Molecular weight
99.131
clogP
0.849
clogS
-0.193
Frequency
0.0041
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
20.31
Number of Rings
1
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
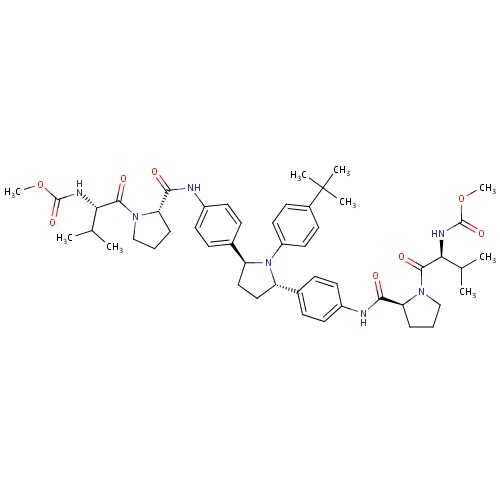
| FDBD01793 | Ombitasvir |
|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; | For use in combination with paritaprevir, ritonavir and dasabuvir for the treatment of HCV genotype 1, and with paritaprevir and ritonavir for the treatment of HCV genotype 4. |
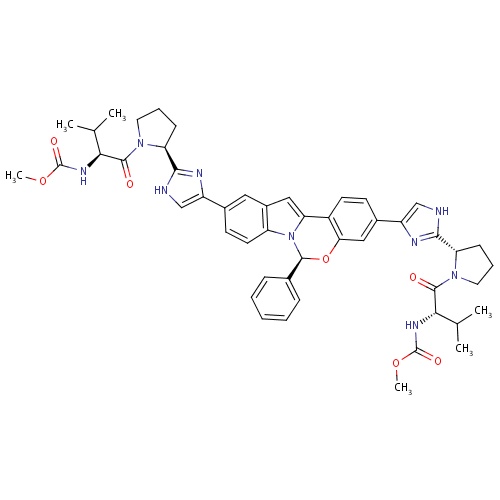
| FDBD01836 | Elbasvir |
|
; |
12 ,
2
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3ddu_ligand_frag_1.mol2 | 3ddu | 1 | -6.35 | C(=O)N1CCCC1 | 7 |
| 4bcd_ligand_frag_7.mol2 | 4bcd | 1 | -6.34 | C(=O)N1CCCC1 | 7 |
| 2rip_ligand_frag_1.mol2 | 2rip | 1 | -6.33 | C(=O)N1CCCC1 | 7 |
| 3eq7_ligand_frag_4.mol2 | 3eq7 | 1 | -6.33 | C(=O)N1CCCC1 | 7 |
| 3eq9_ligand_frag_7.mol2 | 3eq9 | 1 | -6.30 | C(=O)N1CCCC1 | 7 |
| 4jlg_ligand_frag_4.mol2 | 4jlg | 1 | -6.26 | C(=O)N1CCCC1 | 7 |
| 2ajd_ligand_frag_1.mol2 | 2ajd | 1 | -6.25 | C(=O)N1CCCC1 | 7 |
| 3eq8_ligand_frag_5.mol2 | 3eq8 | 1 | -6.25 | C(=O)N1CCCC1 | 7 |
| 2g5t_ligand_frag_0.mol2 | 2g5t | 1 | -6.22 | C(=O)N1CCCC1 | 7 |
329 ,
33

