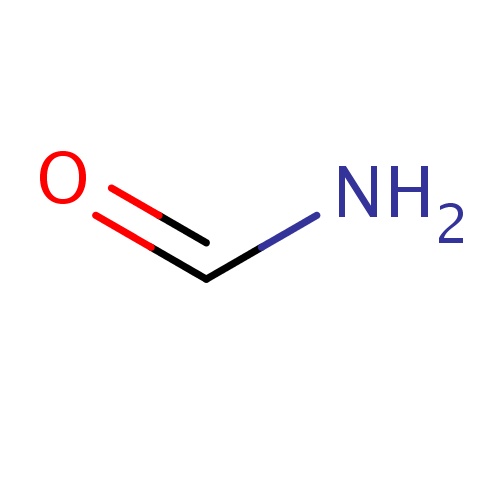
Common name
formamide
IUPAC name
formamide
SMILES
C(=O)N
Common name
formamide
IUPAC name
formamide
SMILES
C(=O)N
INCHI
InChI=1S/CH3NO/c2-1-3/h1H,(H2,2,3)
FORMULA
CH3NO

Common name
formamide
IUPAC name
formamide
Molecular weight
45.041
clogP
-0.474
clogS
0.761
Frequency
0.1240
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
43.09
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00757 | Remifentanil |
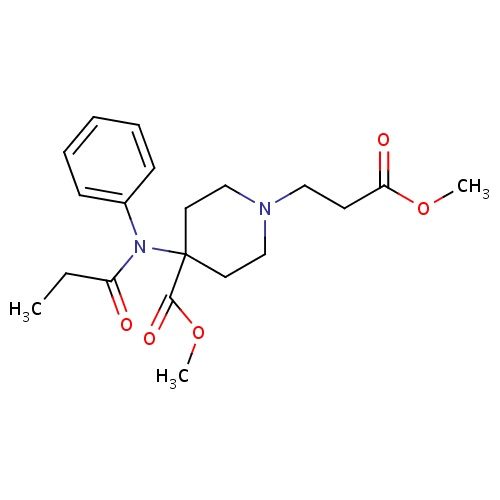
|
Hypnotics and Sedatives; Anesthetics, Intravenous; Analgesics, Opioid; Anesthetics; Nervous System; Anesthetics, General; Opioid Anesthetics; | For use during the induction and maintenance of general anesthesia. |
| FDBD00762 | Bimatoprost |
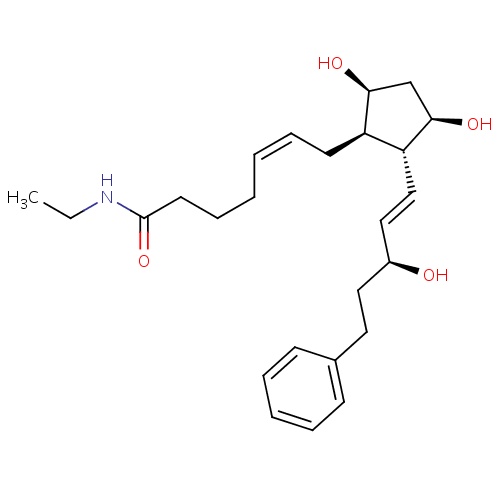
|
Antihypertensive Agents; Anti-glaucoma Agents; Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | For the reduction of elevated intraocular pressure in patients with open angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication. |
| FDBD00769 | Repaglinide |
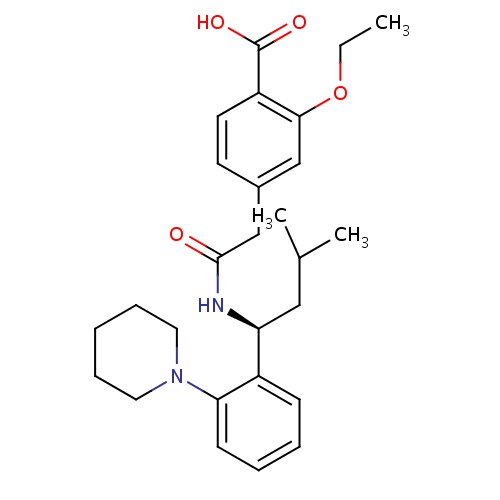
|
Hypoglycemic Agents; Antidiabetic Agents; Meglitinides; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | As an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. |
| FDBD00780 | Ceforanide |
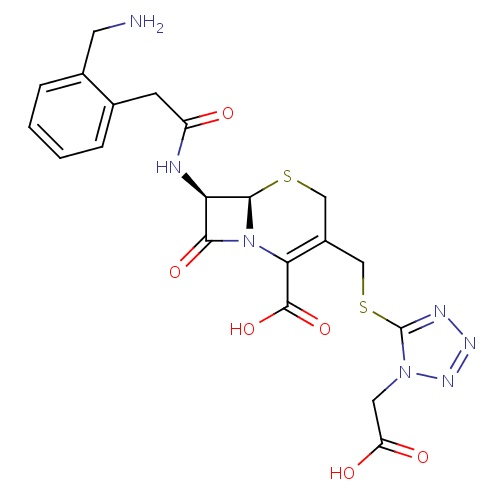
|
Anti-Bacterial Agents; Cephalosporins; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Second-Generation Cephalosporins; | For the treatment of infections caused by susceptible organisms. |
| FDBD00786 | Methacycline |
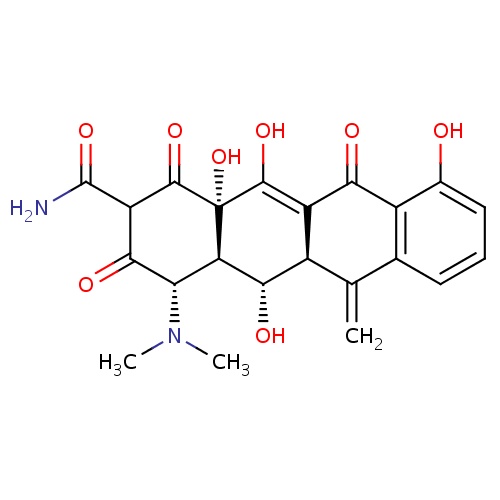
|
Anti-Bacterial Agents; Tetracyclines; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; | For the treatment of acute bacterial exacerbations of chronic bronchitis. |
| FDBD00803 | Mezlocillin |
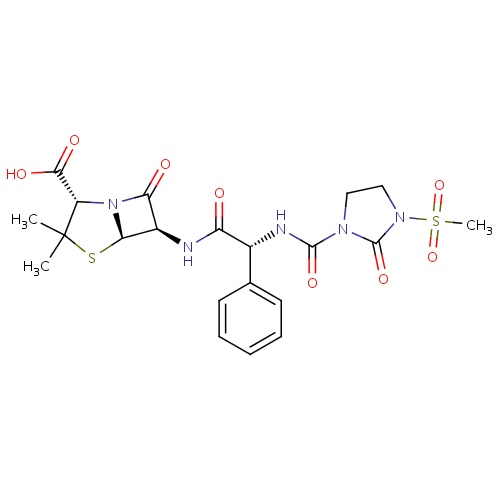
|
Anti-Bacterial Agents; Penicillins; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Beta-Lactam Antibacterials, Penicillins; Penicillins With Extended Spectrum; | Used to treat serious gram–negative infections of the lungs, urinary tract, and skin. |
| FDBD00816 | Mepivacaine |
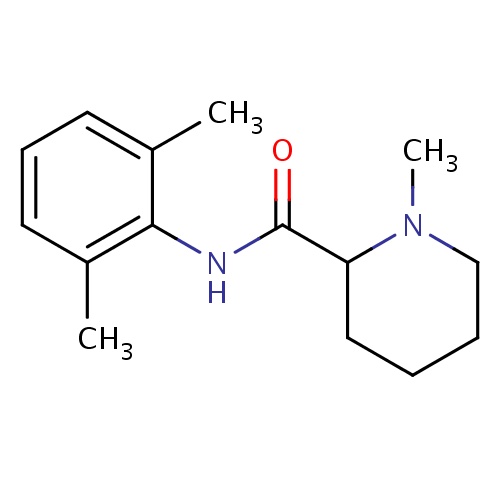
|
Anesthetics, Local; Anesthetics; Nervous System; Amides; | For production of local or regional analgesia and anesthesia by local infiltration, peripheral nerve block techniques, and central neural techniques including epidural and caudal blocks. |
| FDBD00824 | Dactinomycin |
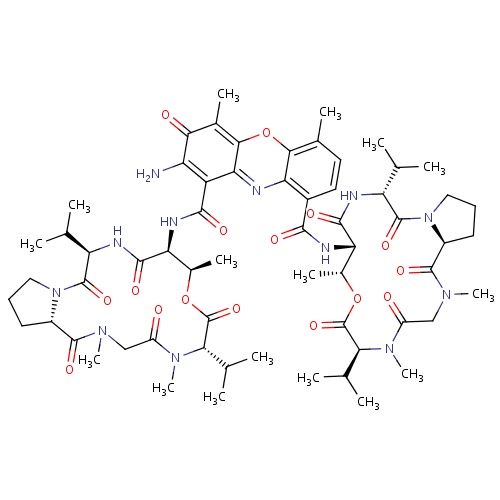
|
Antineoplastic Agents; Immunosuppressive Agents; Anti-Bacterial Agents; Protein Synthesis Inhibitors; Nucleic Acid Synthesis Inhibitors; Antibiotics, Antineoplastic; Antibiotics; Antineoplastic and Immunomodulating Agents; Cytotoxic Antibiotics and Related Substances; Actinomycines; | For the treatment of Wilms' tumor, childhood rhabdomyosarcoma, Ewing's sarcoma and metastatic, nonseminomatous testicular cancer as part of a combination chemotherapy and/or multi-modality treatment regimen. |
| FDBD00834 | Ramelteon |
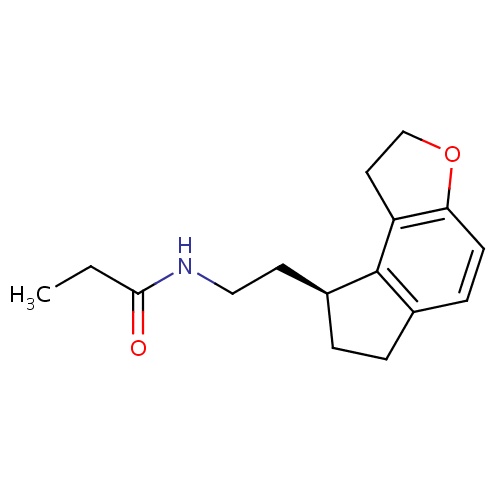
|
Hypnotics and Sedatives; Nervous System; Psycholeptics; Melatonin Receptor Agonists; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | For the treatment of insomnia characterized by difficulty with sleep onset. |
| FDBD00837 | Formoterol |

|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); Beta2 Agonists; | For use as long-term maintenance treatment of asthma in patients 6 years of age and older with reversible obstructive airways disease, including patients with symptoms of nocturnal asthma, who are using optimal corticosteroid treatment and experiencing regular or frequent breakthrough symptoms requiring use of a short-acting bronchodilator. Not indicated for asthma that can be successfully managed with occasional use of an inhaled, short-acting beta2-adrenergic agonist. Also used for the prevention of exercise-induced bronchospasm, as well as long-term treatment of bronchospasm associated with COPD. |
361 ,
37
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2vl1_ligand_frag_1.mol2 | 2vl1 | 1 | -5.59 | C(=O)N | 3 |
| 1wdn_ligand_frag_3.mol2 | 1wdn | 1 | -5.56 | C(=O)N | 3 |
| 1db5_ligand_frag_4.mol2 | 1db5 | 1 | -5.51 | C(=O)N | 3 |
| 2ntf_ligand_frag_10.mol2 | 2ntf | 1 | -5.51 | C(=O)N | 3 |
| 4pml_ligand_frag_1.mol2 | 4pml | 1 | -5.51 | C(=O)N | 3 |
| 4xmb_ligand_frag_8.mol2 | 4xmb | 1 | -5.51 | C(=O)N | 3 |
| 4tjy_ligand_frag_1.mol2 | 4tjy | 1 | -5.50 | C(=O)N | 3 |
| 4f1q_ligand_frag_4.mol2 | 4f1q | 1 | -5.49 | C(=O)N | 3 |
4319 ,
432

