
Common name
indole
IUPAC name
indole
SMILES
[nH]1ccc2c1cccc2
Common name
indole
IUPAC name
indole
SMILES
[nH]1ccc2c1cccc2
INCHI
InChI=1S/C8H7N/c1-2-4-8-7(3-1)5-6-9-8/h1-6,9H
FORMULA
C8H7N

Common name
indole
IUPAC name
indole
Molecular weight
117.148
clogP
2.663
clogS
-2.322
Frequency
0.0086
HBond Acceptor
0
HBond Donor
1
Total PolarSurface Area
15.79
Number of Rings
2
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01484 | Histrelin |
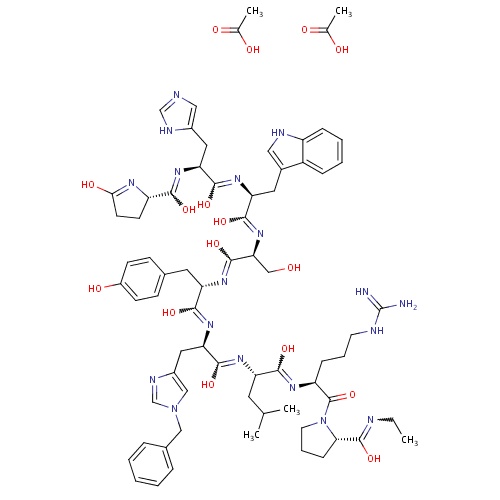
|
Histrelin acetate has two indications | |
| FDBD01507 | Triptorelin |
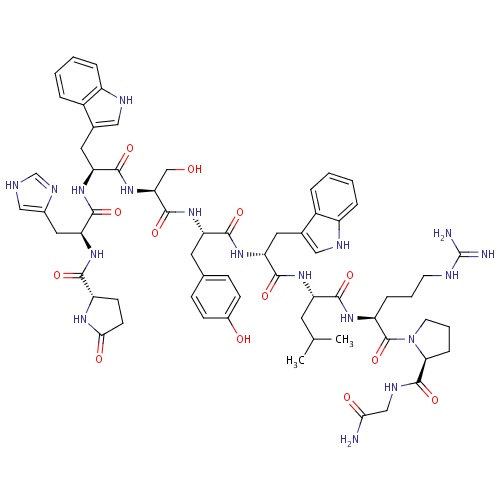
|
Antineoplastic Agents, Hormonal; Luteolytic Agents; Antineoplastic and Immunomodulating Agents; Gonadotropin Releasing Hormone Analogues; Hormones and Related Agents; Endocrine Therapy; Pituitary and Hypothalamic Hormones and Analogues; Systemic Hormonal Preparations, Excl. Sex Hormones and Insulins; Hypothalamic Hormones; Gonadotropin-Releasing Hormones; | Triptorelin is indicated for the palliative treatment of advanced prostate cancer. |
| FDBD01680 | Somatostatin |
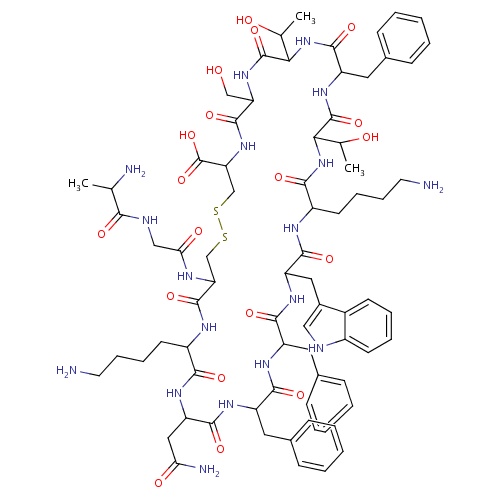
|
Hormones; Pituitary and Hypothalamic Hormones and Analogues; Systemic Hormonal Preparations, Excl. Sex Hormones and Insulins; Hypothalamic Hormones; Somatostatin and Analogues; | For the symptomatic treatment of acute bleeding from esophageal varices. Other treatment options for long-term management of the condition may be considered if necessary, once initial control has been established. |
| FDBD01709 | Sincalide |
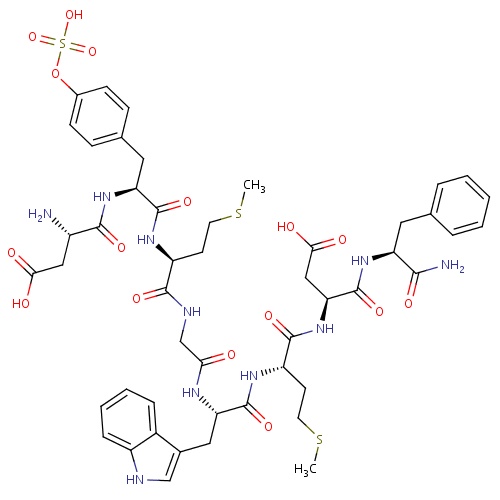
|
Diagnostic Agents; Tests for Bile Duct Patency; | |
| FDBD01769 | Lixisenatide |

|
Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; | For lowering blood glucose level in patients with type 2 diabetes. |
25 ,
3
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2vuk_ligand_1_1.mol2 | 2vuk | 1 | -8.20 | C1CCC[C@H]2[C@H]3CCCC[C@H]3[N@H+]([C@@H]12)CC | 15 |
| 1tow_ligand_2_2.mol2 | 1tow | 1 | -8.13 | CC[N@@H+]1[C@H]2CCCC[C@@H]2[C@@H]2[C@H]1CCCC2 | 15 |
| 1tow_ligand_1_2.mol2 | 1tow | 1 | -7.89 | [N@H+]1([C@H]2CCCC[C@@H]2[C@@H]2[C@H]1CCCC2)C | 14 |
| 1tow_ligand_frag_3.mol2 | 1tow | 1 | -7.81 | [NH2+]1[C@H]2CCCC[C@@H]2[C@@H]2[C@H]1CCCC2 | 13 |
| 4us3_ligand_1_1.mol2 | 4us3 | 1 | -7.79 | [C@@H]1(C[NH2+][C@H]2[C@H]1CCCC2)C | 10 |
| 2pql_ligand_1_1.mol2 | 2pql | 1 | -7.75 | C[C@@H]1C[NH2+][C@@H]2[C@H]1CCCC2 | 10 |
| 4us4_ligand_1_1.mol2 | 4us4 | 1 | -7.73 | C[C@H]1C[NH2+][C@H]2[C@@H]1CCCC2 | 10 |
| 4zim_ligand_frag_0.mol2 | 4zim | 1 | -7.71 | C1CC[C@@H]2[C@H](C1)[C@H]1[C@H](CCCC1)[NH2+]2 | 13 |
| 2vuk_ligand_frag_0.mol2 | 2vuk | 1 | -7.70 | C1CCC[C@H]2[C@H]3CCCC[C@H]3[NH2+][C@@H]12 | 13 |
| 3dxk_ligand_2_0.mol2 | 3dxk | 1 | -7.69 | CC[C@@H]1[C@@H](C)[NH2+][C@H]2CCCC[C@@H]12 | 12 |
489 ,
49

