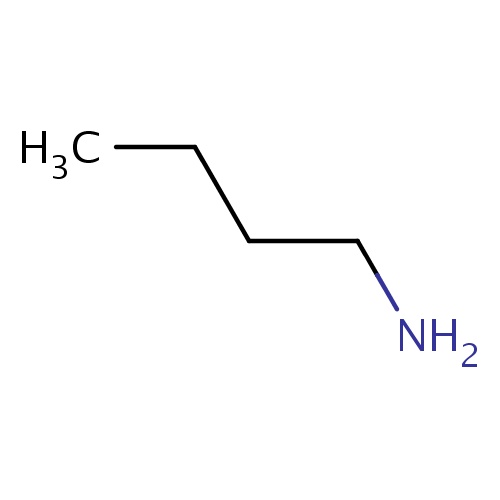
Common name
butan-1-amine
IUPAC name
butan-1-amine
SMILES
C(CCN)C
Common name
butan-1-amine
IUPAC name
butan-1-amine
SMILES
C(CCN)C
INCHI
InChI=1S/C4H11N/c1-2-3-4-5/h2-5H2,1H3
FORMULA
C4H11N

Common name
butan-1-amine
IUPAC name
butan-1-amine
Molecular weight
73.137
clogP
0.198
clogS
-0.986
Frequency
0.0106
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00753 | Benzylpenicilloyl Polylysine |
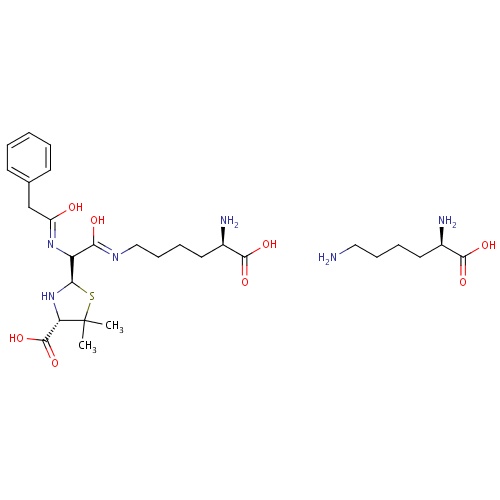
|
Acetamides; | For use as a adjunct in assessing the risk of administering penicillin (benzylpenicillin or penicillin G). |
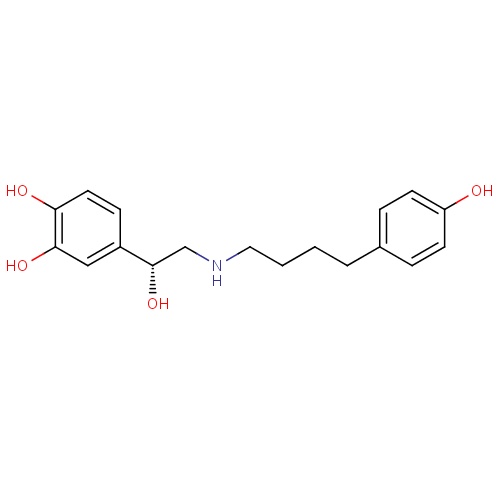
| FDBD00793 | Salmeterol |
|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Beta2 Agonists; | For the treatment of asthma and chronic obstructive pulmonary disease (COPD). |
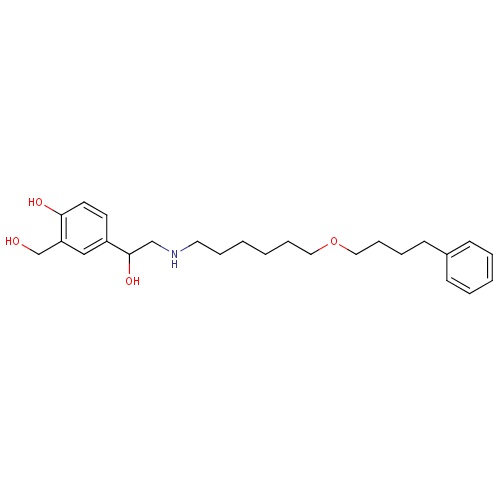
| FDBD00952 | Arbutamine |
|
Cardiovascular System; Cardiac Therapy; Adrenergic and Dopaminergic Agents; Cardiac Stimulants Excl. Cardiac Glycosides; Beta2 Agonists; | Used to elicit acute cardiovascular responses (cardiac stumulant), similar to those produced by exercise, in order to aid in diagnosing the presence or absence of coronary artery disease (CAD) in patients who cannot exercise adequately. |
| FDBD01064 | Halofantrine |
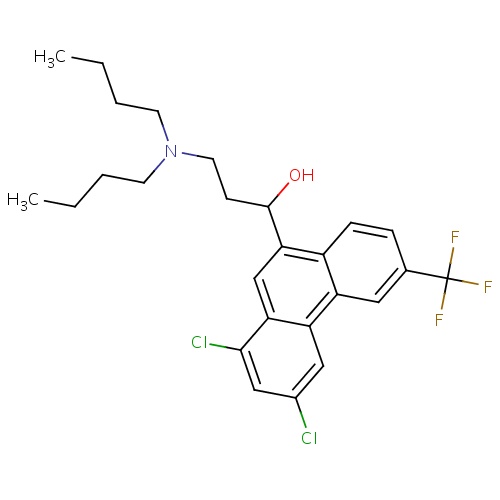
|
Antimalarials; Antiprotozoal Agents; Antiparasitic Products, Insecticides and Repellents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For treatment of Severe malaria. |
| FDBD01099 | Lisdexamfetamine |

|
Central Nervous System Stimulants; Dopamine Uptake Inhibitors; Adrenergic alpha-1 Receptor Antagonists; Nervous System; Psychoanaleptics; Centrally Acting Sympathomimetics; Psychostimulants, Agents Used for Adhd and Nootropics; | For the treatment of Attention Deficit/Hyperactivity Disorder (ADHD) in pediatric populations aged 6 to 12 years. |
| FDBD01127 | Fosamprenavir |
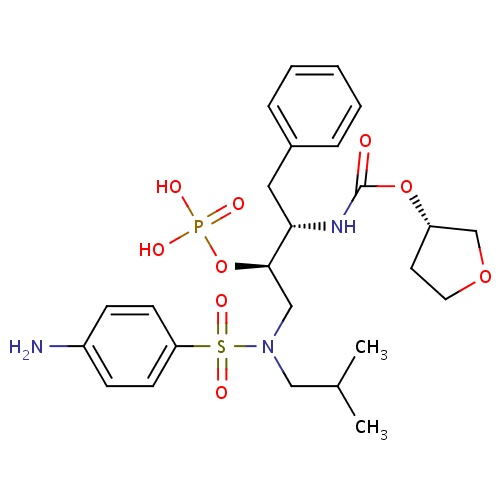
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Prodrugs; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; | Indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. The use of fosamprenavir is pending revision due to a potential association between the drug and myocardial infarction and dyslipidemia in HIV infected adults. |
| FDBD01333 | Dronedarone |
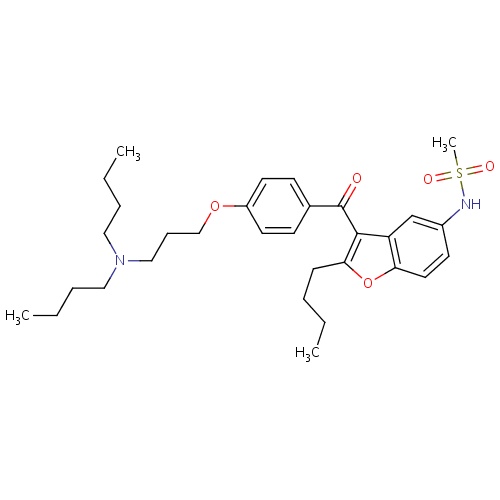
|
Anti-Arrhythmia Agents; Adrenergic alpha-1 Receptor Antagonists; Cardiovascular System; Antiarrhythmics, Class III; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Management of paroxysmal or persistent atrial fibrillation via restoration of normal sinus rhythm. |
| FDBD01343 | Vapreotide |
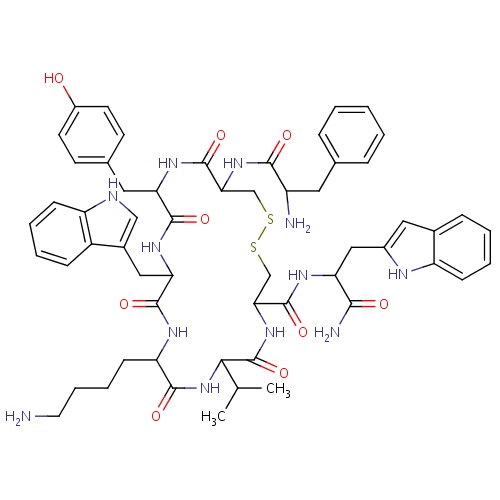
|
Antineoplastic Agents; Analgesics; Pituitary and Hypothalamic Hormones and Analogues; Systemic Hormonal Preparations, Excl. Sex Hormones and Insulins; Hypothalamic Hormones; Somatostatin and Analogues; | For the treatment of esophageal variceal bleeding in patients with cirrhotic liver disease and has also shown efficacy in the treatment of patients with AIDS-related diarrhea. |
| FDBD01429 | Pasireotide |
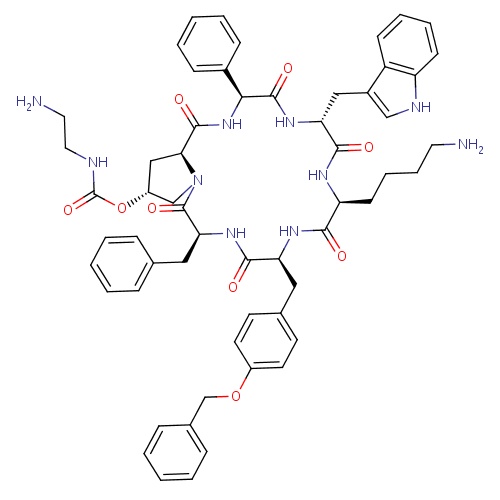
|
Pituitary and Hypothalamic Hormones and Analogues; Systemic Hormonal Preparations, Excl. Sex Hormones and Insulins; Hypothalamic Hormones; Somatostatin and Analogues; | For the treatment of Cushing's disease, specifically for those patients whom pituitary surgery has not been curative or is not an option. |
| FDBD01448 | Lumefantrine |
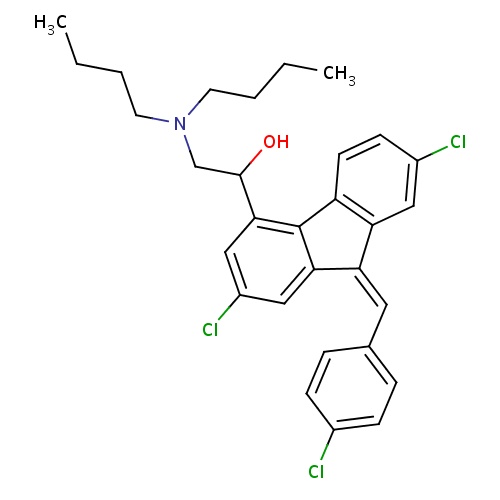
|
Antimalarials; Antiprotozoal Agents; Antiparasitic Products, Insecticides and Repellents; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Lumefantrine and artemether combination therapy is indicated for the treatment of acute uncomplicated malaria caused by . |
31 ,
4
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4q4i_ligand_2_1.mol2 | 4q4i | 1 | -6.52 | [NH3+]CCC(C)C | 6 |
| 3g19_ligand_2_9.mol2 | 3g19 | 1 | -6.50 | [NH3+]CCC(C)C | 6 |
| 3b3c_ligand_2_1.mol2 | 3b3c | 1 | -6.38 | C(C[NH3+])C(C)C | 6 |
| 3b7i_ligand_2_1.mol2 | 3b7i | 1 | -6.30 | C(C)(C)CC[NH3+] | 6 |
| 2v16_ligand_3_100.mol2 | 2v16 | 1 | -6.29 | C([NH3+])CC(C)(C)C | 7 |
| 4kx8_ligand_2_1.mol2 | 4kx8 | 1 | -6.28 | C(C)(C)CC[NH3+] | 6 |
| 2pvu_ligand_3_3.mol2 | 2pvu | 1 | -6.22 | CCCC[NH3+] | 5 |
| 1ft7_ligand_2_1.mol2 | 1ft7 | 1 | -6.20 | C(C)(C)CC[NH3+] | 6 |
| 1tmn_ligand_2_1.mol2 | 1tmn | 1 | -6.20 | C(C(C)C)C[NH3+] | 6 |
295 ,
30

