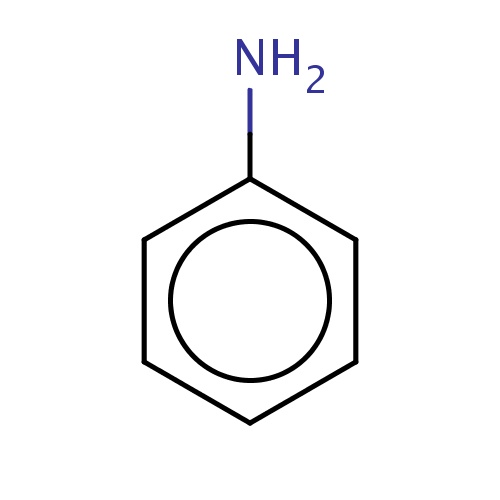
Common name
aniline
IUPAC name
aniline
SMILES
Nc1ccccc1
Common name
aniline
IUPAC name
aniline
SMILES
Nc1ccccc1
INCHI
InChI=1S/C6H7N/c7-6-4-2-1-3-5-6/h1-5H,7H2
FORMULA
C6H7N

Common name
aniline
IUPAC name
aniline
Molecular weight
93.126
clogP
1.260
clogS
-1.189
Frequency
0.0247
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01603 | Floctafenine |
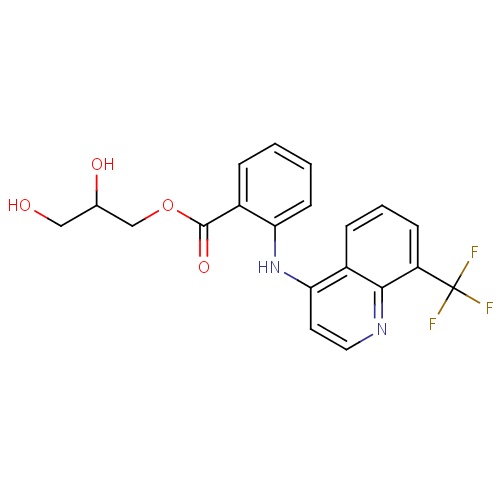
|
Analgesics; Nervous System; | |
| FDBD01608 | Etofenamate |
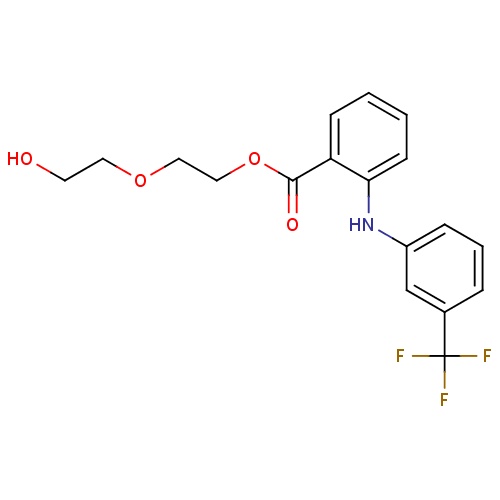
|
Musculo-Skeletal System; Antiinflammatory Preparations, Non-Steroids for Topical Use; Topical Products for Joint and Muscular Pain; | |
| FDBD01651 | Ceritinib |
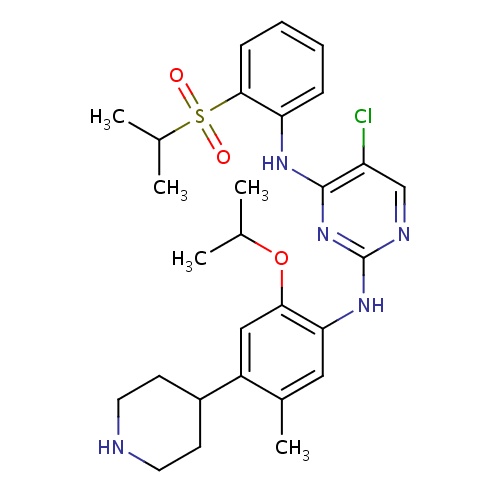
|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; | Ceritinib is a kinase inhibitor indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib. This indication is approved under accelerated approval based on tumor response rate and duration of response. An improvement in survival or disease-related symptoms has not been established. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials. |
| FDBD01662 | Nintedanib |
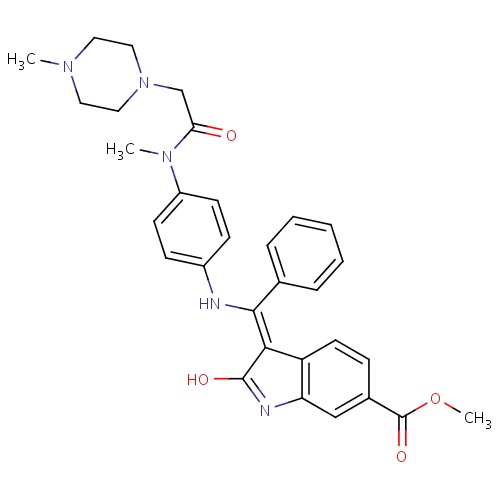
|
Antineoplastic Agents; Enzyme Inhibitors; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; | Nintedanib is indicated for the treatment of idiopathic pulmonary fibrosis (IPF). |
| FDBD01668 | Tetracaine |
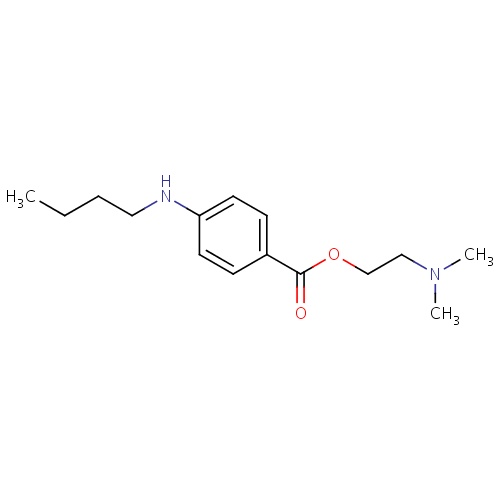
|
Anesthetics, Local; Anesthetics; Ophthalmologicals; Sensory Organs; Dermatologicals; Nervous System; Cardiovascular System; Local Anesthetics; Agents for Treatment of Hemorrhoids and Anal Fissures for Topical Use; Vasoprotectives; Anesthetics for Topical Use; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Esters of Aminobenzoic Acid; | |
| FDBD01705 | Isosulfan blue |
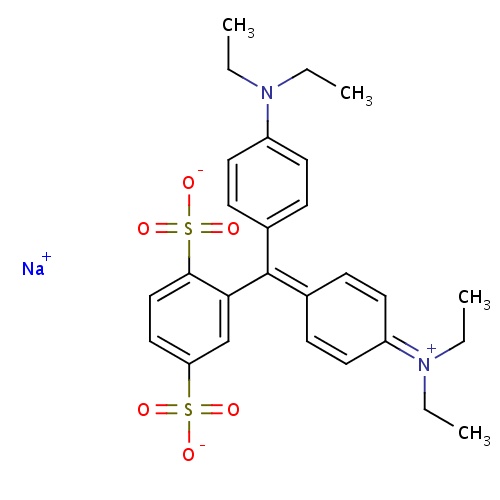
|
; | |
| FDBD01759 | Efonidipine |
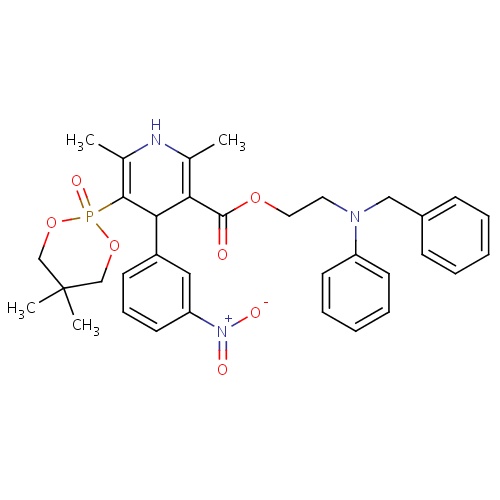
|
; | For the treatment of hypertension. |
| FDBD01783 | Morniflumate |
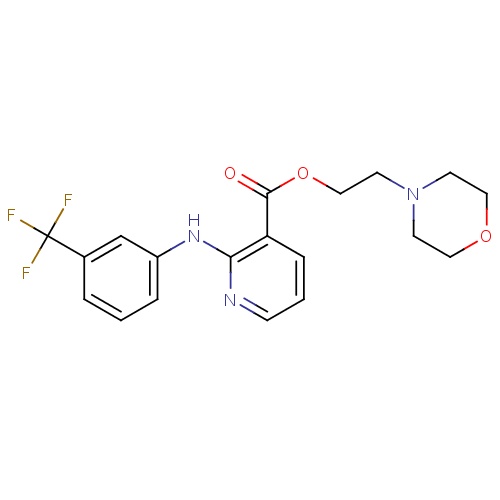
|
Musculo-Skeletal System; Antiinflammatory and Antirheumatic Products, Non-Steroids; Antiinflammatory and Antirheumatic Products; | |
| FDBD01792 | Talniflumate |
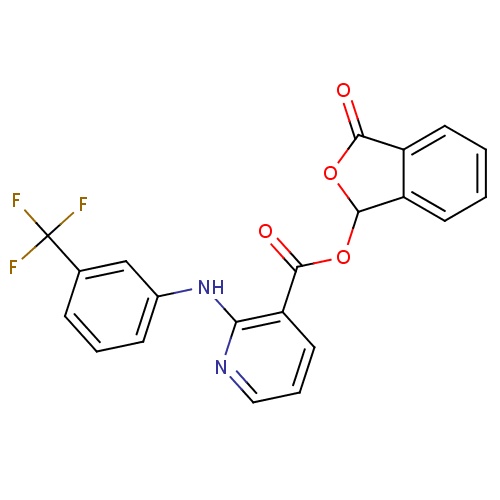
|
; | For the treatment of cystic fibrosis. |
| FDBD01810 | Osimertinib |
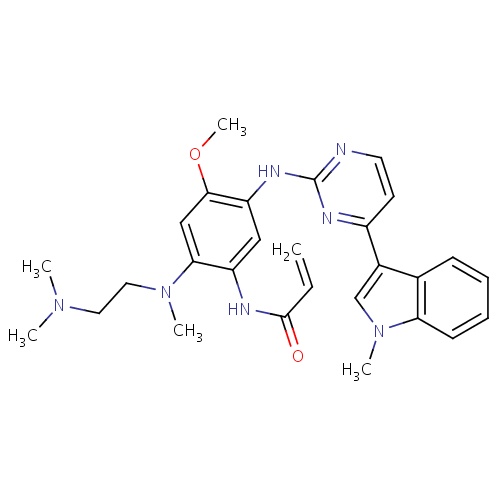
|
Antineoplastic Agents; Protein Kinase Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | Osimertinib is indicated for the treatment of patients with metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer (NSCLC), as detected by an FDA- approved test, who have progressed on or after EGFR-TKI therapy. |
72 ,
8
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3fui_ligand_1_3.mol2 | 3fui | 1 | -6.80 | c1ccc(cc1)N | 7 |
| 4l02_ligand_1_2.mol2 | 4l02 | 1 | -6.77 | Nc1ccccc1 | 7 |
| 4o2a_ligand_frag_2.mol2 | 4o2a | 1 | -6.76 | c1(ccccc1)N | 7 |
| 3ftw_ligand_1_0.mol2 | 3ftw | 1 | -6.75 | c1ccccc1N | 7 |
| 2vba_ligand_1_1.mol2 | 2vba | 1 | -6.74 | c1(ccccc1)N | 7 |
| 3ftv_ligand_1_0.mol2 | 3ftv | 1 | -6.74 | c1ccccc1N | 7 |
| 3ftz_ligand_frag_0.mol2 | 3ftz | 1 | -6.74 | Nc1ccccc1 | 7 |
| 1cx9_ligand_frag_0.mol2 | 1cx9 | 1 | -6.72 | c1cc(ccc1)N | 7 |
| 2hz0_ligand_1_6.mol2 | 2hz0 | 1 | -6.72 | c1ccccc1N | 7 |
| 4p72_ligand_1_3.mol2 | 4p72 | 1 | -6.71 | Nc1ccccc1 | 7 |
542 ,
55

