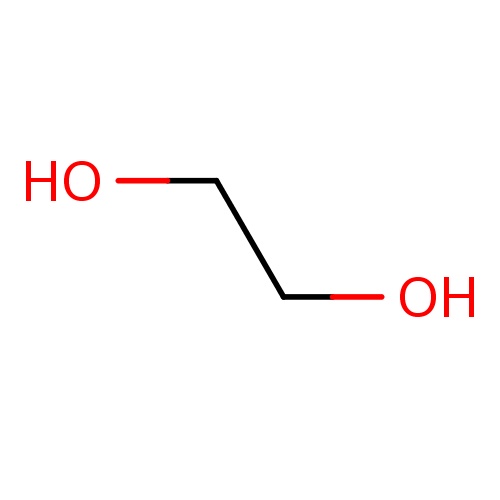
Common name
ethylene glycol
IUPAC name
ethylene glycol
SMILES
C(O)CO
Common name
ethylene glycol
IUPAC name
ethylene glycol
SMILES
C(O)CO
INCHI
InChI=1S/C2H6O2/c3-1-2-4/h3-4H,1-2H2
FORMULA
C2H6O2

Common name
ethylene glycol
IUPAC name
ethylene glycol
Molecular weight
62.068
clogP
-0.637
clogS
0.629
Frequency
0.0265
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
40.46
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01350 | Ospemifene |

|
Estrogen Antagonists; Selective Estrogen Receptor Modulators; Sex Hormones and Modulators of the Genital System; Genito Urinary System and Sex Hormones; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; | Ospemifene is used for the treatment of moderate to dyspareunia, a symptom of vulvar and vaginal atrophy, due to menopause. |
| FDBD01370 | Sodium stibogluconate |

|
Antineoplastic Agents; Antiprotozoal Agents; Schistosomicides; Antiparasitic Products, Insecticides and Repellents; Agents Against Protozoal Diseases; Agents Against Leishmaniasis and Trypanosomiasis; Antimony Compounds; | For the treatment of various types of a protozoal infection called leishmaniasis, which may result from sandfly bites in tropical and temperate parts of the world. Also investigated for use/treatment in cancer/tumors (unspecified) and solid tumors. |
| FDBD01416 | Tyloxapol |

|
Surface-Active Agents; Expectorants; Detergents; Cough and Cold Preparations; Respiratory System; | Used as a surfactant to aid liquefaction and removal of mucopurulent (containing mucus and pus) bronchopulmonary secretions, administered by inhalation through a nebulizer or with a stream of oxygen. Also investigated for use/treatment in cystic fibrosis and pediatric indications. |
| FDBD01443 | Gadobutrol |
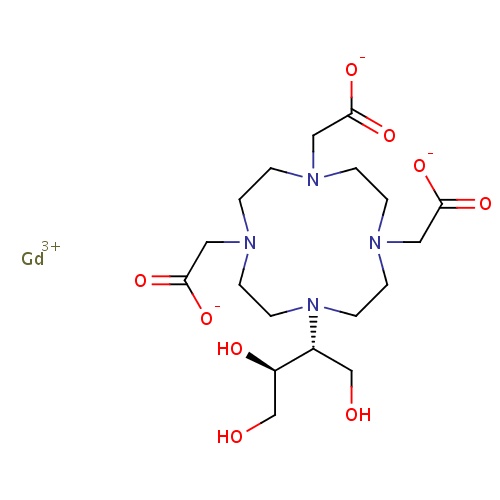
|
Contrast Media; Paramagnetic Contrast Media; Magnetic Resonance Imaging Contrast Media; | For diagnostic use only. Indicated for adults and children age 2 and over for contrast enhancement during cranial and spinal MRI, and for contrast-enhanced magnetic resonance angiography (CE-MRA). Gadobutrol is particularly suited for the detection of very small lesions and for the visualization of tumors that do not readily take up contrast media. It may be a desired agent when the exclusion or demonstration of an additional pathology may influence the choice of therapy or patient management. It may also be suitable for perfusion studies in the diagnosis of stroke, detection of focal cerebral ischemia, and in studies of tumor perfusion. |
| FDBD01525 | Bupranolol |
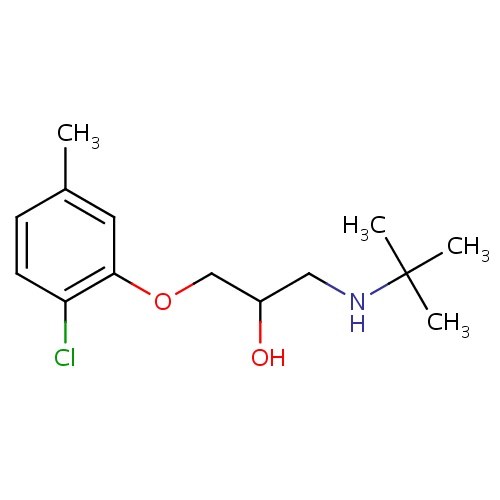
|
Antihypertensive Agents; Anti-Arrhythmia Agents; Adrenergic beta-Antagonists; Cardiovascular System; Beta Blocking Agents; Beta Blocking Agents, Non-Selective; | Used to manage hypertension and tachycardia. Also used to treat glaucoma. |
| FDBD01529 | Ticagrelor |

|
Platelet Aggregation Inhibitors; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Purinergic P2Y Receptor Antagonists; CYP3A4 Inhibitors; | For the prevention of thrombotic events (for example stroke or heart attack) in patients with acute coronary syndrome or myocardial infarction with ST elevation. |
| FDBD01582 | MACITENTAN |
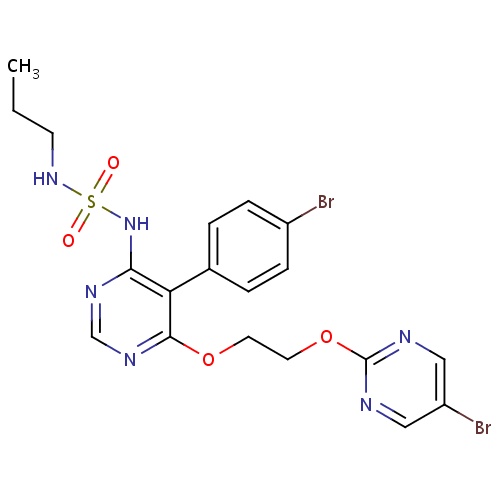
|
Antihypertensive Agents; Vasodilator Agents; Cardiovascular System; Antihypertensives for Pulmonary Arterial Hypertension; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; Endothelin A Receptor Antagonists; Endothelin B Receptor Antagonists; | Macitentan is indicated for patients with pulmonary arterial hypertension. |
| FDBD01603 | Floctafenine |
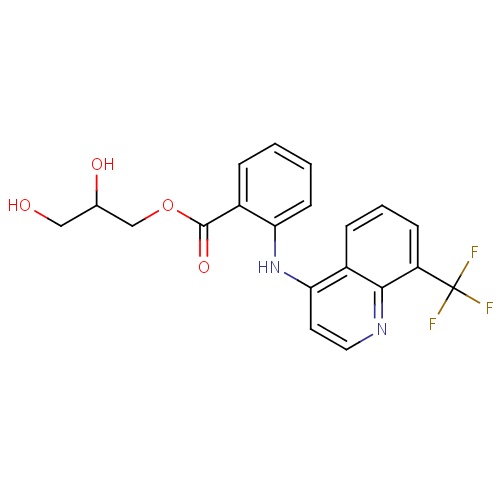
|
Analgesics; Nervous System; | |
| FDBD01608 | Etofenamate |
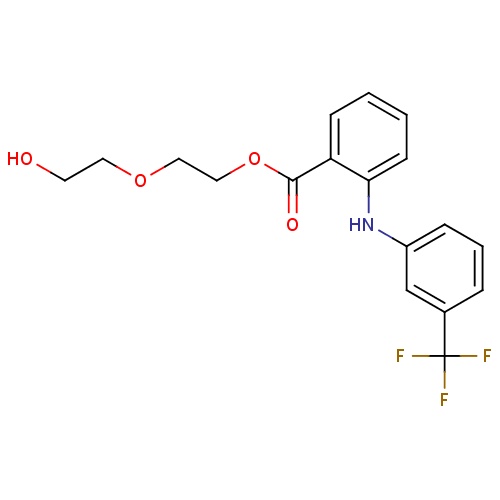
|
Musculo-Skeletal System; Antiinflammatory Preparations, Non-Steroids for Topical Use; Topical Products for Joint and Muscular Pain; | |
| FDBD01665 | Vilanterol |
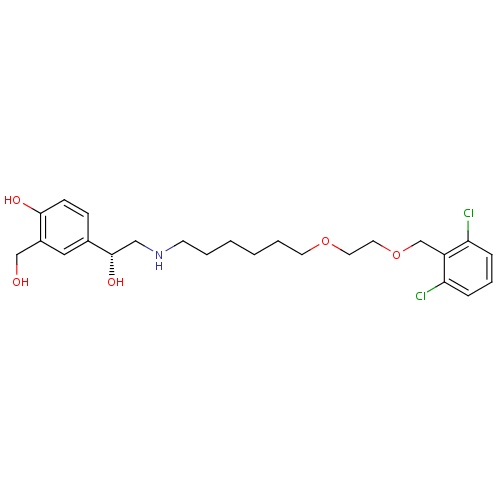
|
Immunosuppressive Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Adrenergics, Inhalants; CYP3A4 Inhibitors; Beta2 Agonists; | Vilanterol is approved for use in several combination products such as with fluticasone furoate under the tradename Breo Ellipta and in combination with umeclidinium bromide as Anoro Ellipta. Approved by the FDA in 2013, use of Breo Ellipta is indicated for the long-term, once-daily maintenance treatment of airflow obstruction in patients with COPD, including chronic bronchitis and emphysema. It is also indicated for once-daily maintenance treatment of asthma in patients aged 18 or older with reversible obstructive airways disease. |
77 ,
8

