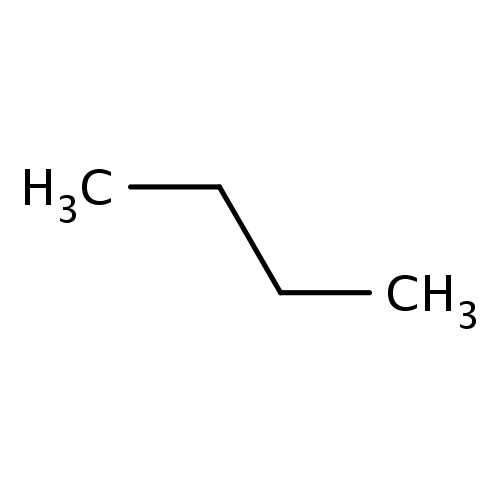
Common name
butane
IUPAC name
butane
SMILES
CCCC
Common name
butane
IUPAC name
butane
SMILES
CCCC
INCHI
InChI=1S/C4H10/c1-3-4-2/h3-4H2,1-2H3
FORMULA
C4H10

Common name
butane
IUPAC name
butane
Molecular weight
58.122
clogP
0.893
clogS
-1.283
Frequency
0.0680
HBond Acceptor
0
HBond Donor
0
Total PolarSurface Area
0
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00746 | Bumetanide |
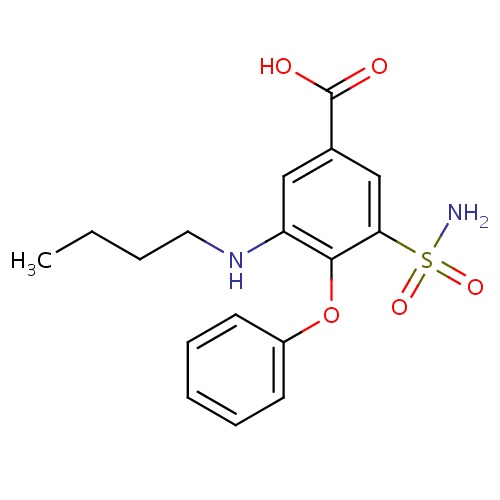
|
Diuretics; Sodium Potassium Chloride Symporter Inhibitors; Cardiovascular System; Sulfonamides, Plain; High-Ceiling Diuretics; High-Ceiling Diuretics and Potassium-Sparing Agents; | For the treatment of edema associated with congestive heart failure, hepatic and renal disease including the nephrotic syndrome. |
| FDBD00751 | Oxybuprocaine |
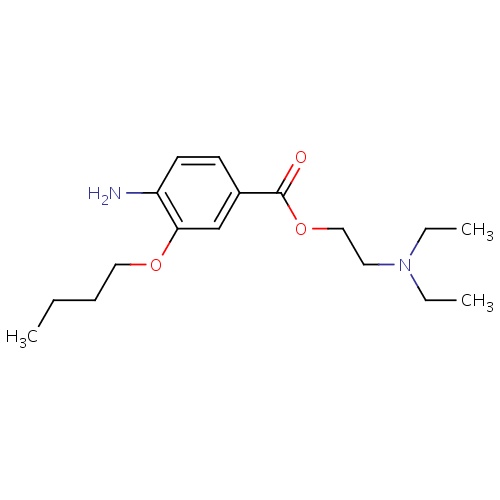
|
Anesthetics, Local; Ophthalmologicals; Sensory Organs; Dermatologicals; Local Anesthetics; Anesthetics for Topical Use; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; | Used to temporarily numb the front surface of the eye so that the eye pressure can be measured or a foreign body removed. |
| FDBD00753 | Benzylpenicilloyl Polylysine |
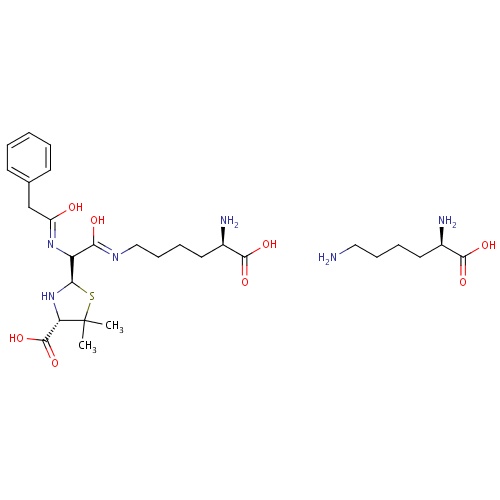
|
Acetamides; | For use as a adjunct in assessing the risk of administering penicillin (benzylpenicillin or penicillin G). |
| FDBD00774 | Dinoprostone |
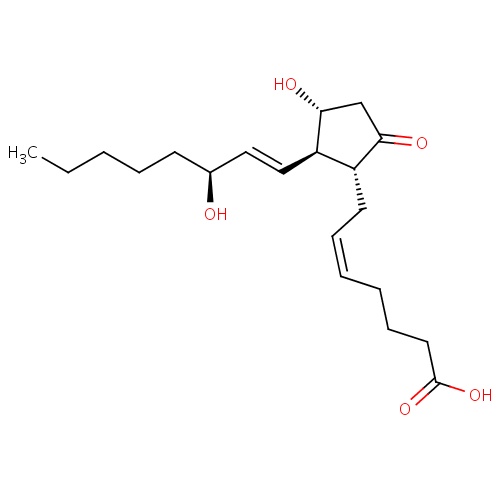
|
For the termination of pregnancy during the second trimester (from the 12th through the 20th gestational week as calculated from the first day of the last normal menstrual period), as well as for evacuation of the uterine contents in the management of missed abortion or intrauterine fetal death up to 28 weeks of gestational age as calculated from the first day of the last normal menstrual period. Also used in the management of nonmetastatic gestational trophoblastic disease (benign hydatidiform mole). Other indications include improving the cervical inducibility (cervical ; | |
| FDBD00785 | Misoprostol |
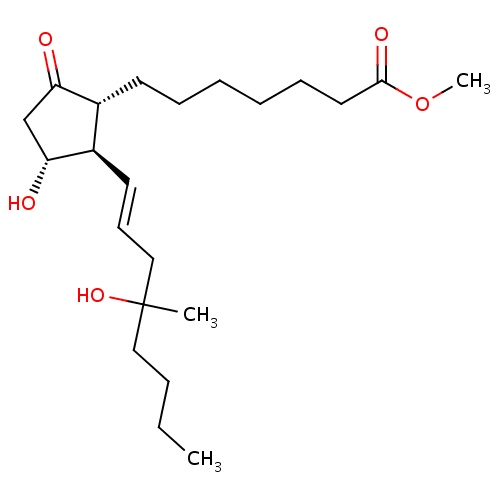
|
Indicated for the treatment of ulceration (duodenal, gastric and NSAID induced) and prophylaxis for NSAID induced ulceration. | |
| FDBD00793 | Salmeterol |
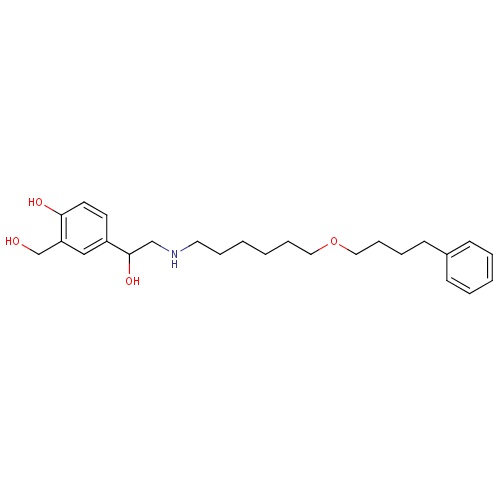
|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Beta2 Agonists; | For the treatment of asthma and chronic obstructive pulmonary disease (COPD). |
| FDBD00796 | Hexafluronium |
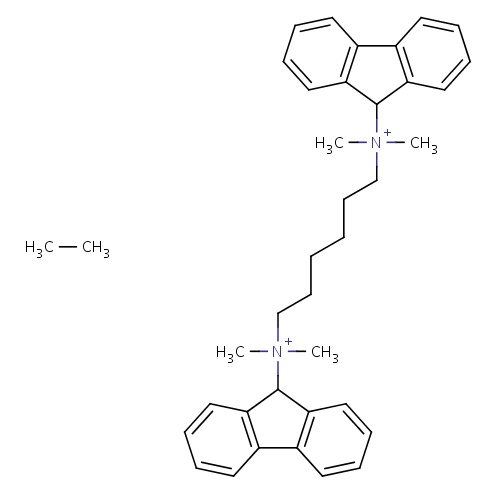
|
Neuromuscular Agents; Musculo-Skeletal System; Muscle Relaxants; Muscle Relaxants, Peripherally Acting Agents; | Used as an adjunct with succinylcholine (or suxamethonium chloride) to prolong muscle relaxation and to prevent succinylcholine-induced muscle fasciculations. |
| FDBD00799 | Demecarium |
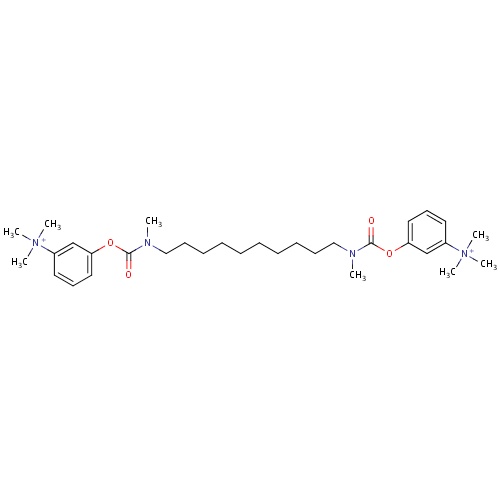
|
Parasympathomimetics; Cholinergic Agents; Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; | For the topical treatment of chronic open-angle glaucoma. |
| FDBD00802 | Fulvestrant |

|
Antineoplastic Agents; Antineoplastic Agents, Hormonal; Estrogen Antagonists; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Anti-Estrogens; CYP3A4 Inhibitors; Estrogen Receptor Antagonists; | For the treatment of hormone receptor positive metastatic breast cancer in postmenopausal women with disease progression following anti-estrogen therapy. |
| FDBD00830 | Telithromycin |
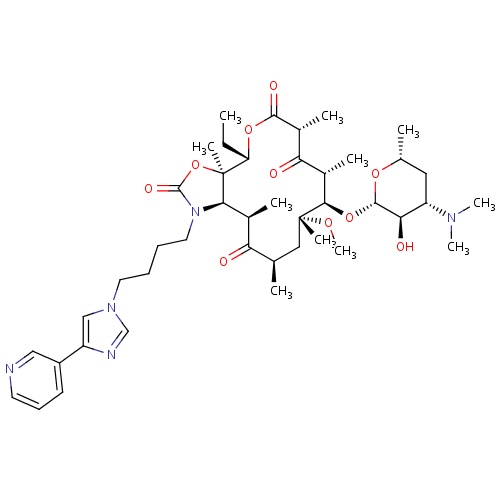
|
Anti-Bacterial Agents; Ketolides; Macrolides; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Macrolides, Lincosamides and Streptogramins; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of . |
198 ,
20
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4m3f_ligand_2_7.mol2 | 4m3f | 1 | -6.83 | C(C(C)C)C | 5 |
| 3cvk_ligand_2_5.mol2 | 3cvk | 1 | -6.80 | C(C(C)(C)C)C | 6 |
| 1yt7_ligand_2_51.mol2 | 1yt7 | 1 | -6.59 | C(C(C)(C)C)C | 6 |
| 1gt3_ligand_2_0.mol2 | 1gt3 | 1 | -6.53 | CC(C)CC | 5 |
| 2v12_ligand_3_651.mol2 | 2v12 | 1 | -6.50 | C(C)(C)C(C)C | 6 |
| 2v0z_ligand_3_749.mol2 | 2v0z | 1 | -6.49 | CC(C)C(C)C | 6 |
| 4u82_ligand_3_251.mol2 | 4u82 | 1 | -6.46 | C(C(C)C)C | 5 |
| 4djh_ligand_2_9.mol2 | 4djh | 1 | -6.45 | C(C(C)C)C | 5 |
| 1dmt_ligand_2_26.mol2 | 1dmt | 1 | -6.44 | C(C(C)C)C | 5 |
| 1fbm_ligand_3_16.mol2 | 1fbm | 1 | -6.44 | C(C)(C)CC | 5 |
2388 ,
239

