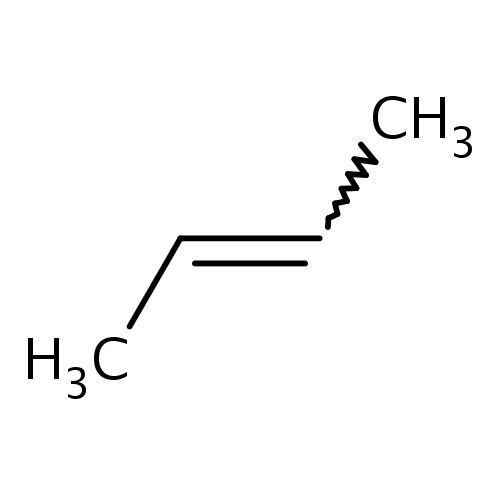
Common name
(Z)-but-2-ene
IUPAC name
(Z)-but-2-ene
SMILES
CC=CC
Common name
(Z)-but-2-ene
IUPAC name
(Z)-but-2-ene
SMILES
CC=CC
INCHI
InChI=1S/C4H8/c1-3-4-2/h3-4H,1-2H3/b4-3-
FORMULA
C4H8

Common name
(Z)-but-2-ene
IUPAC name
(Z)-but-2-ene
Molecular weight
56.106
clogP
0.714
clogS
-0.556
Frequency
0.0069
HBond Acceptor
0
HBond Donor
0
Total PolarSurface Area
0
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00024 | Alpha-Linolenic Acid |
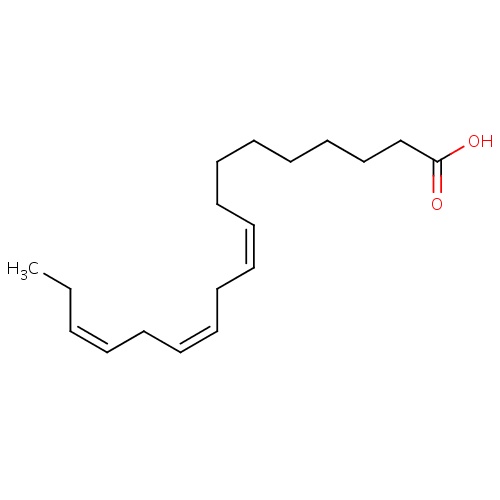
|
Dietary Supplements; Micronutrients; Supplements; | For nutritional supplementation and for treating dietary shortage or imbalance. |
| FDBD00045 | Dihomo-gamma-linolenic acid |
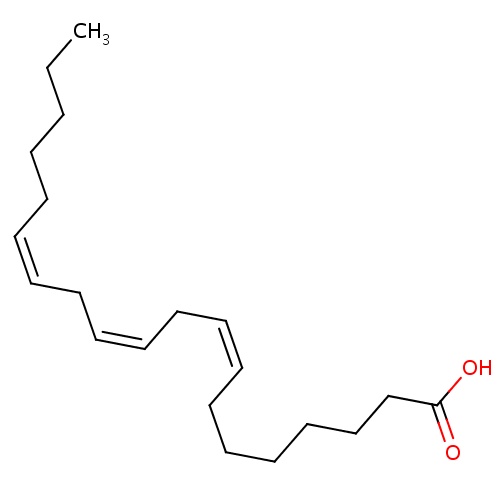
|
Dietary Supplements; Micronutrients; Supplements; | For nutritional supplementation, also for treating dietary shortage or imbalance. |
| FDBD00049 | Icosapent |
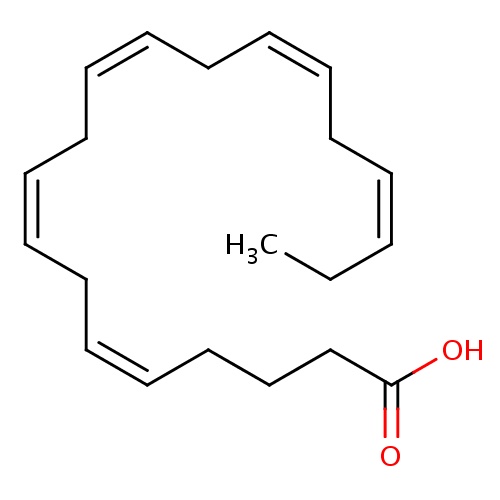
|
Dietary Supplements; Micronutrients; Supplements; Omega-3 Fatty Acids; | EPA can be used for lowering elevated triglycerides in those who are hyperglyceridemic. In addition, EPA may play a therapeutic role in patients with cystic fibrosis by reducing disease severity and may play a similar role in type 2 diabetics in slowing the progression of diabetic nephropathy. |
| FDBD00174 | Travoprost |
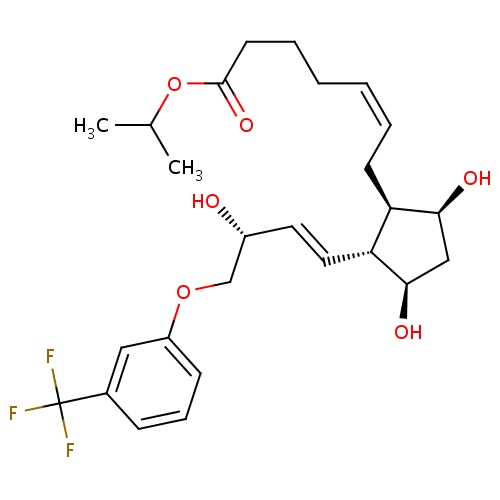
|
Antihypertensive Agents; Prostaglandins, Synthetic; Anti-glaucoma Agents; Ophthalmics; Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | Ophthalmic solution used for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication. |
| FDBD00306 | Carboprost Tromethamine |
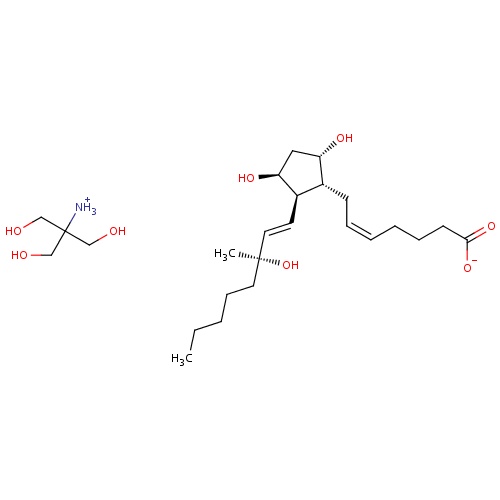
|
Oxytocics; Abortifacient Agents, Nonsteroidal; Prostaglandins; Genito Urinary System and Sex Hormones; Uterotonics; Tromethamine; | For aborting pregnancy between the 13th and 20th weeks of gestation as calculated from the first day of the last normal menstrual period and in the following conditions related to second trimester abortion: 1. Failure of expulsion of the fetus during the course of treatment by another method; 2. Premature rupture of membranes in intrauterine methods with loss of drug and insufficient or absent uterine activity; 3. Requirement of a repeat intrauterine instillation of drug for expulsion of the fetus; 4. Inadvertent or spontaneous rupture of membranes in the presence of a previable fetus and absence of adequate activity for expulsion. Also for the treatment of postpartum hemorrhage due to uterine atony which has not responded to conventional methods of management. |
| FDBD00519 | Latanoprost |
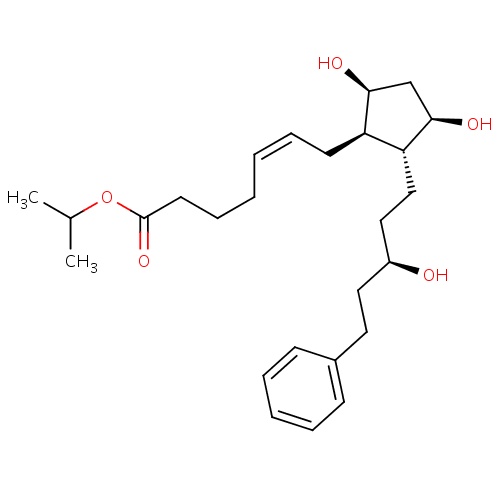
|
Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | For the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension. |
| FDBD00553 | Mycophenolate mofetil |
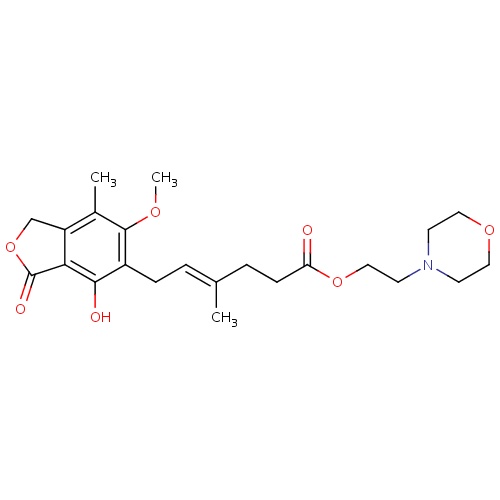
|
Immunosuppressive Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | For the prophylaxis of organ rejection in patients receiving allogeneic renal, cardiac or hepatic transplants. Mycophenolate mofetil should be used concomitantly with cyclosporine and corticosteroids. |
| FDBD00762 | Bimatoprost |
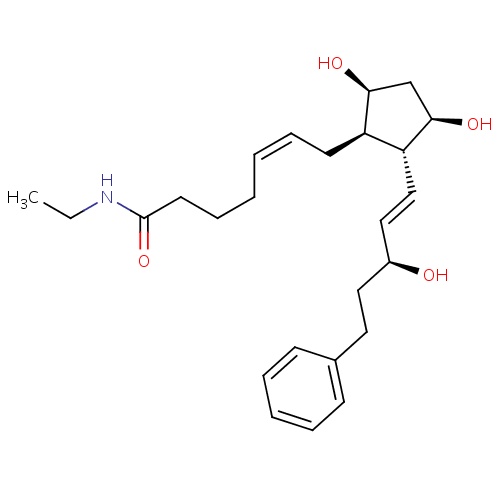
|
Antihypertensive Agents; Anti-glaucoma Agents; Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | For the reduction of elevated intraocular pressure in patients with open angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication. |
| FDBD00774 | Dinoprostone |
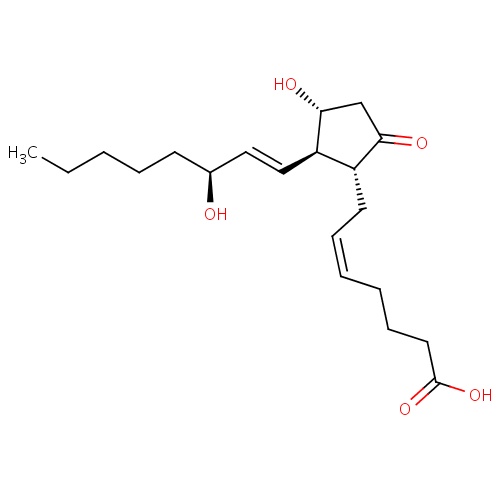
|
For the termination of pregnancy during the second trimester (from the 12th through the 20th gestational week as calculated from the first day of the last normal menstrual period), as well as for evacuation of the uterine contents in the management of missed abortion or intrauterine fetal death up to 28 weeks of gestational age as calculated from the first day of the last normal menstrual period. Also used in the management of nonmetastatic gestational trophoblastic disease (benign hydatidiform mole). Other indications include improving the cervical inducibility (cervical ; | |
| FDBD00876 | Phylloquinone |
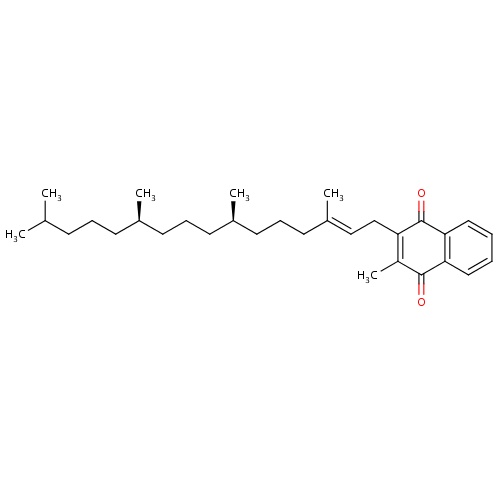
|
Vitamins; Antifibrinolytic Agents; Vitamin K; Blood and Blood Forming Organs; Antihemorrhagics; | For the treatment of haemorrhagic conditions in infants, antidote for coumarin anticoagulants in hypoprothrombinaemia. |
20 ,
3
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4jhq_ligand_3_394.mol2 | 4jhq | 1 | -6.37 | C/C=C/C | 4 |
| 4prg_ligand_2_81.mol2 | 4prg | 1 | -6.34 | C(=C\C)/C | 4 |
| 2chm_ligand_2_35.mol2 | 2chm | 1 | -6.29 | C(=C\C)/C | 4 |
| 3dzt_ligand_2_84.mol2 | 3dzt | 1 | -6.28 | C/C=C\C | 4 |
| 5d1w_ligand_3_66.mol2 | 5d1w | 1 | -6.23 | C(=C\C)/C | 4 |
138 ,
14

