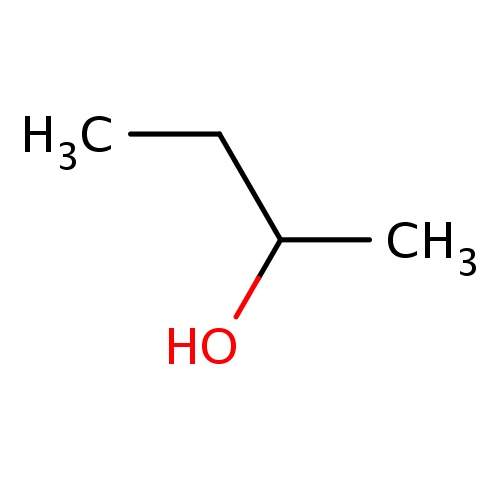
Common name
(2S)-butan-2-ol
IUPAC name
(2S)-butan-2-ol
SMILES
CC(O)CC
Common name
(2S)-butan-2-ol
IUPAC name
(2S)-butan-2-ol
SMILES
CC(O)CC
INCHI
InChI=1S/C4H10O/c1-3-4(2)5/h4-5H,3H2,1-2H3/t4-/m0/s1
FORMULA
C4H10O

Common name
(2S)-butan-2-ol
IUPAC name
(2S)-butan-2-ol
Molecular weight
74.122
clogP
0.266
clogS
-0.399
Frequency
0.0034
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
20.23
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
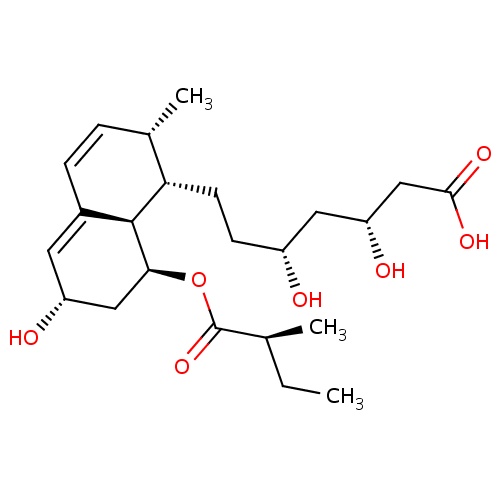
| FDBD00064 | Pravastatin |
|
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of hypercholesterolemia and to reduce the risk of cardiovascular disease. |
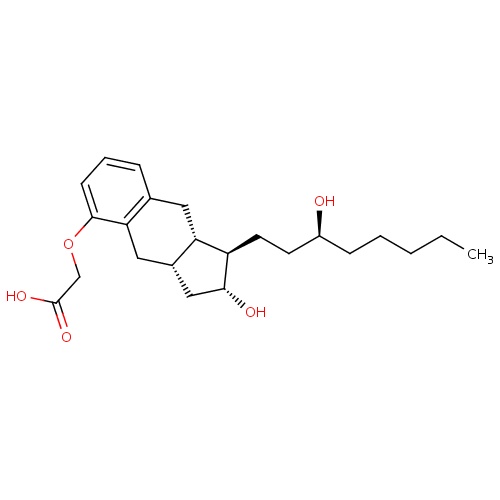
| FDBD00256 | Treprostinil |
|
Antithrombins; Anticoagulants; Antihypertensive Agents; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; | For use as a continuous subcutaneous infusion or intravenous infusion (for those not able to tolerate a subcutaneous infusion) for the treatment of pulmonary arterial hypertension in patients with NYHA Class II-IV symptoms to diminish symptoms associated with exercise. |
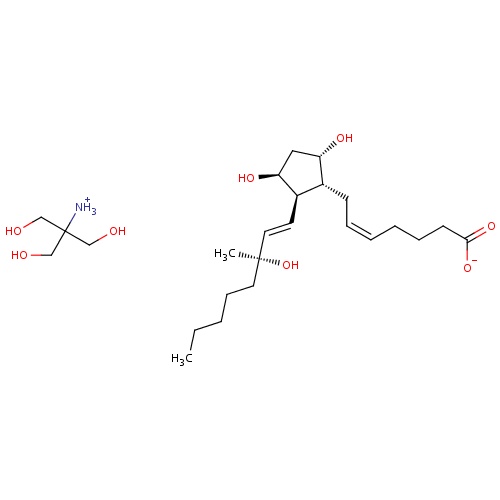
| FDBD00306 | Carboprost Tromethamine |
|
Oxytocics; Abortifacient Agents, Nonsteroidal; Prostaglandins; Genito Urinary System and Sex Hormones; Uterotonics; Tromethamine; | For aborting pregnancy between the 13th and 20th weeks of gestation as calculated from the first day of the last normal menstrual period and in the following conditions related to second trimester abortion: 1. Failure of expulsion of the fetus during the course of treatment by another method; 2. Premature rupture of membranes in intrauterine methods with loss of drug and insufficient or absent uterine activity; 3. Requirement of a repeat intrauterine instillation of drug for expulsion of the fetus; 4. Inadvertent or spontaneous rupture of membranes in the presence of a previable fetus and absence of adequate activity for expulsion. Also for the treatment of postpartum hemorrhage due to uterine atony which has not responded to conventional methods of management. |
| FDBD00376 | Ritonavir |

|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
| FDBD00519 | Latanoprost |
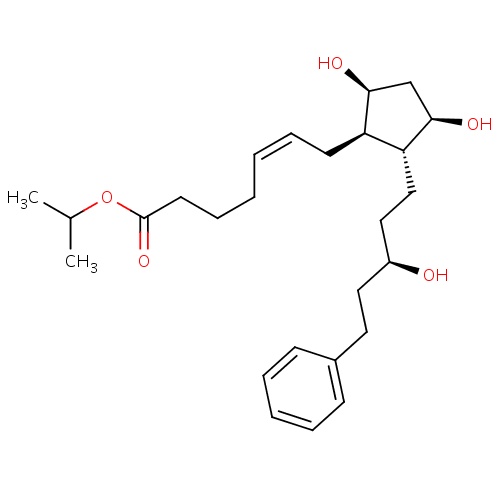
|
Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | For the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension. |
| FDBD00923 | Atazanavir |
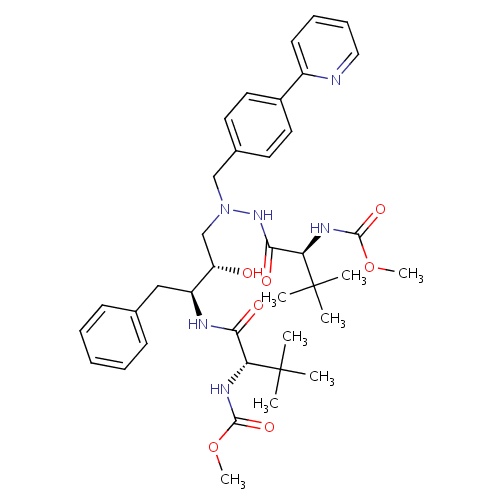
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Used in combination with other antiretroviral agents for the treatment of HIV-1 infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. |
| FDBD00927 | Atorvastatin |
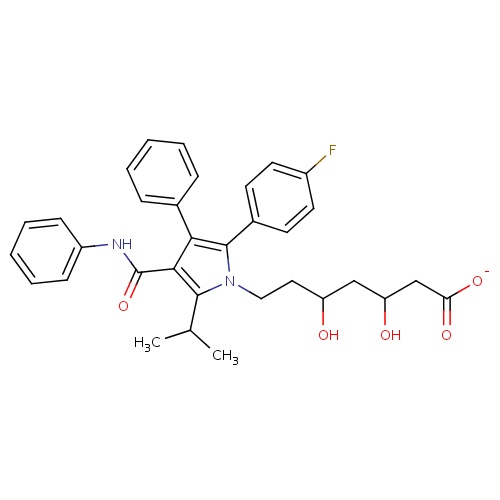
|
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; Dipeptidyl-Peptidase IV Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | |
| FDBD01251 | Lopinavir |
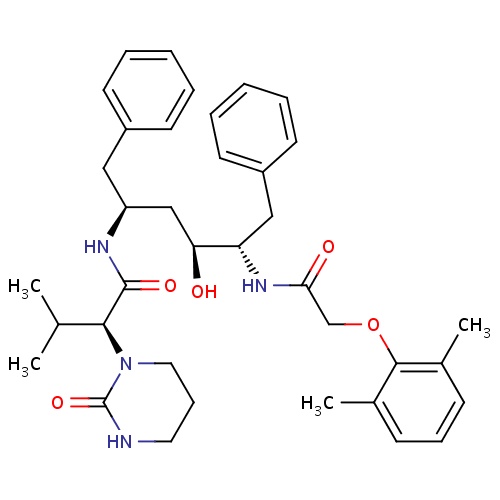
|
Anti-HIV Agents; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
| FDBD03050 | cyproconazole |
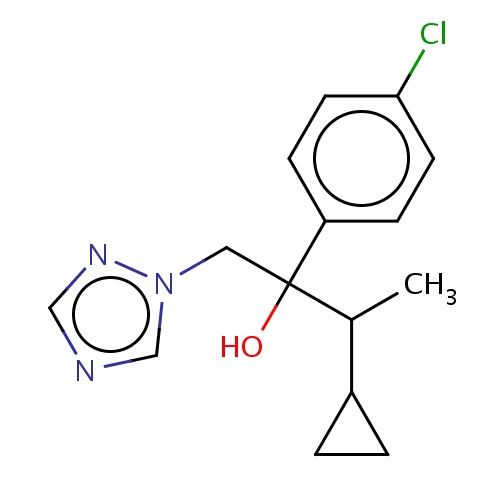
|
Fungicide | Fungicide |
| FDBD03063 | hexaconazole |
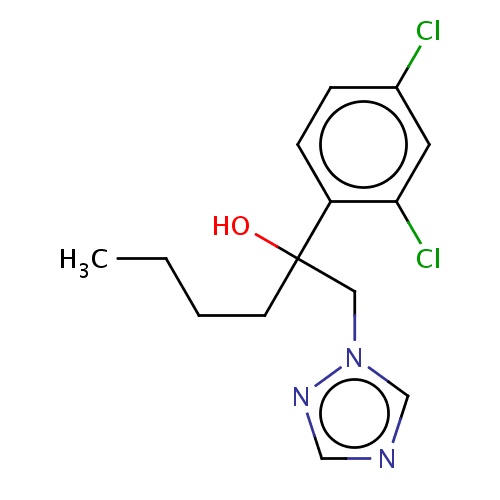
|
Fungicide | Fungicide |
10 ,
2
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4fhi_ligand_2_53.mol2 | 4fhi | 1 | -6.89 | [C@H](O)(C)C(C)(C)C | 7 |
| 4fhh_ligand_2_53.mol2 | 4fhh | 1 | -6.83 | [C@H](O)(C(C)(C)C)C | 7 |
| 1gt3_ligand_1_3.mol2 | 1gt3 | 1 | -6.56 | C(C(O)(C)C)C | 6 |
| 1rjk_ligand_1_3.mol2 | 1rjk | 1 | -6.51 | CCC(O)(C)C | 6 |
| 2o4j_ligand_2_0.mol2 | 2o4j | 1 | -6.50 | CCC(O)(C)C | 6 |
| 2o4r_ligand_1_2.mol2 | 2o4r | 1 | -6.49 | C(C(O)(C)C)C | 6 |
| 1ie9_ligand_1_3.mol2 | 1ie9 | 1 | -6.43 | CCC(O)(C)C | 6 |
| 1db1_ligand_1_3.mol2 | 1db1 | 1 | -6.40 | C(C(O)(C)C)C | 6 |
378 ,
38

