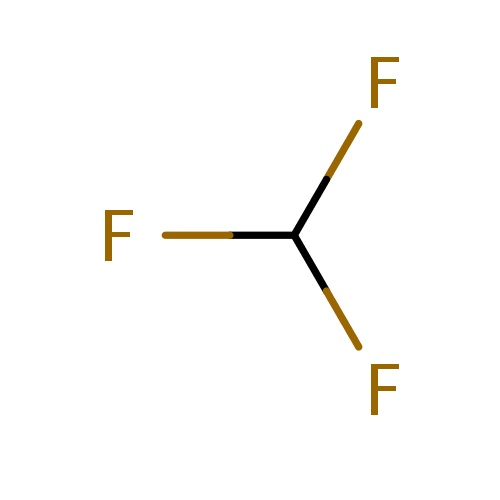
Common name
fluoroform
IUPAC name
fluoroform
SMILES
FC(F)F
Common name
fluoroform
IUPAC name
fluoroform
SMILES
FC(F)F
INCHI
InChI=1S/CHF3/c2-1(3)4/h1H
FORMULA
CHF3

Common name
fluoroform
IUPAC name
fluoroform
Molecular weight
70.014
clogP
1.935
clogS
-0.621
Frequency
0.0704
HBond Acceptor
0
HBond Donor
0
Total PolarSurface Area
0
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
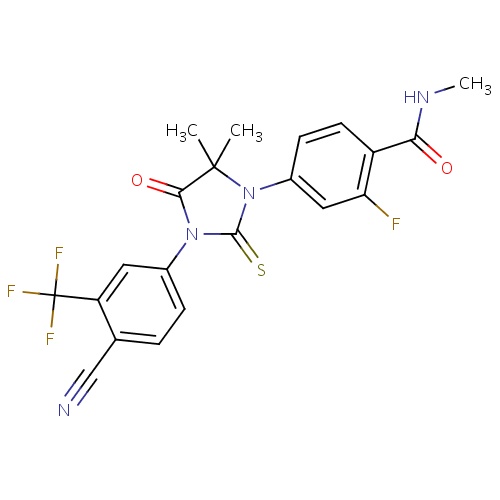
| FDBD01563 | Enzalutamide |
|
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; Endocrine Therapy; Hormone Antagonists and Related Agents; Anti-Androgens; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Enzalutamide is indicated for the treatment of patients with metastatic castration-resistant prostate cancer who have previously received docetaxel. |
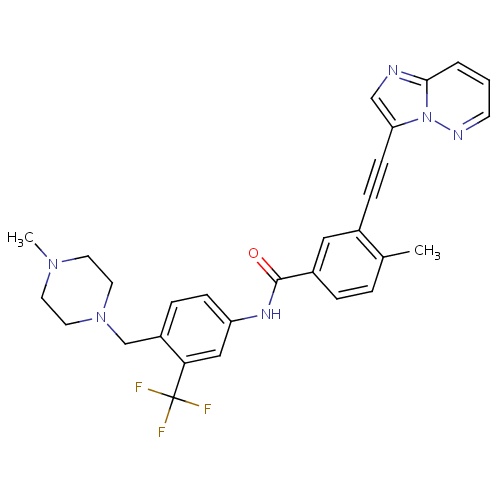
| FDBD01564 | Ponatinib |
|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; BSEP/ABCB11 Inhibitors; | Ponatinib is indicated for the treatment of adult patients with chronic phase, accelerated phase, or blast phase chronic myeloid leukemia (CML) that is resistant or intolerant to prior tyrosine kinase inhibitor therapy or Philadelphia chromosome positive acute lymphoblastic leukemia (Ph+ALL) that is resistant or intolerant to prior tyrosine kinase inhibitor therapy. |
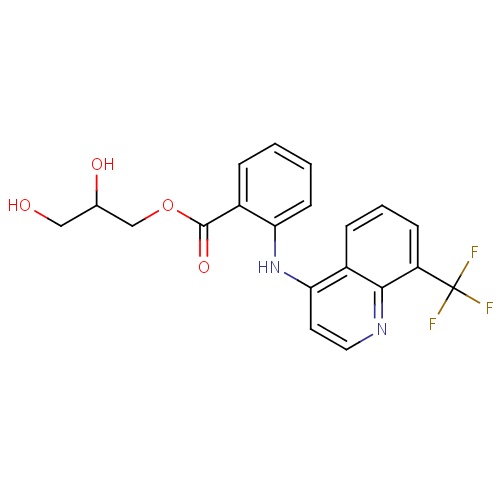
| FDBD01603 | Floctafenine |
|
Analgesics; Nervous System; | |
| FDBD01608 | Etofenamate |
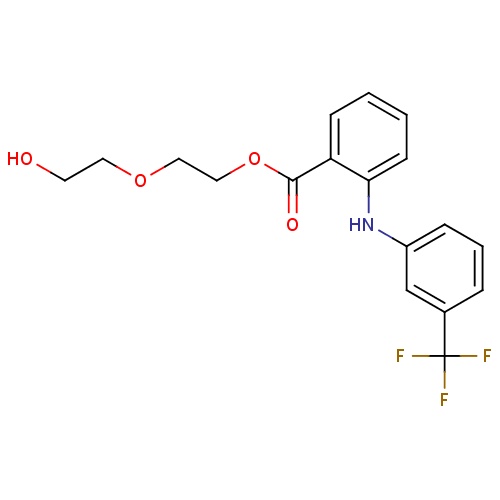
|
Musculo-Skeletal System; Antiinflammatory Preparations, Non-Steroids for Topical Use; Topical Products for Joint and Muscular Pain; | |
| FDBD01645 | Netupitant |
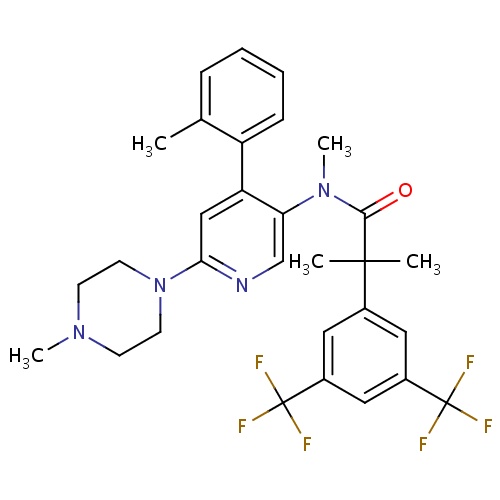
|
Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); | Netupitant is an antiemitic drug approved by the FDA in October 2014 for use in combination with palonosetron for the prevention of acute and delayed vomiting and nausea associated with cancer chemotherapy including highly emetogenic chemotherapy. |
| FDBD01710 | Sonidegib |
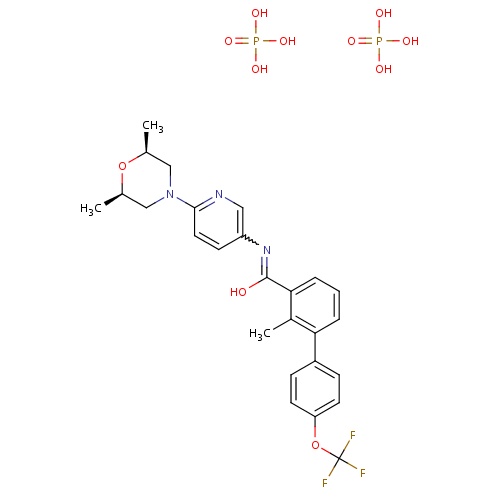
|
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; CYP3A4 Inhibitors; | Sonidegib is approved for use in the US and EU for treatment of adults with locally advanced basal cell carcinoma (BCC) that has recurred post surgery or radiation therapy. It is also approved for adult patients with BCC who are not eligible for surgery or radiation therapy. (2). |
| FDBD01734 | Lorpiprazole |
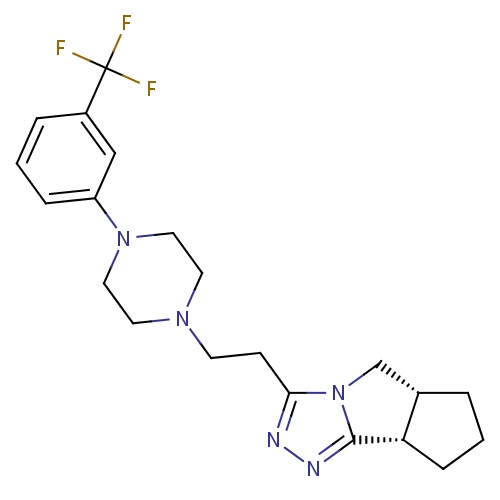
|
; | |
| FDBD01783 | Morniflumate |
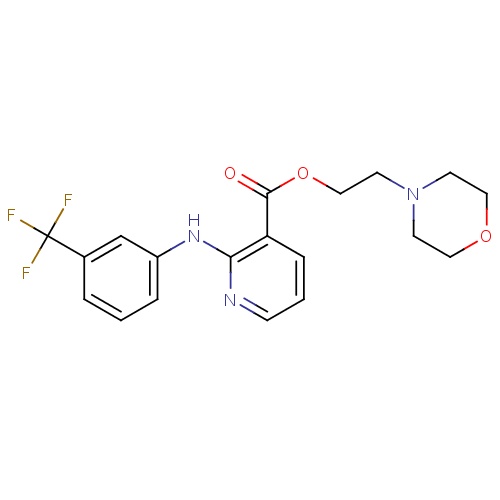
|
Musculo-Skeletal System; Antiinflammatory and Antirheumatic Products, Non-Steroids; Antiinflammatory and Antirheumatic Products; | |
| FDBD01788 | Rolapitant |

|
CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Neurokinin-1 Receptor Antagonists; | Rolapitant is indicated in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including, but not limited to, highly emetogenic chemotherapy. |
| FDBD01792 | Talniflumate |
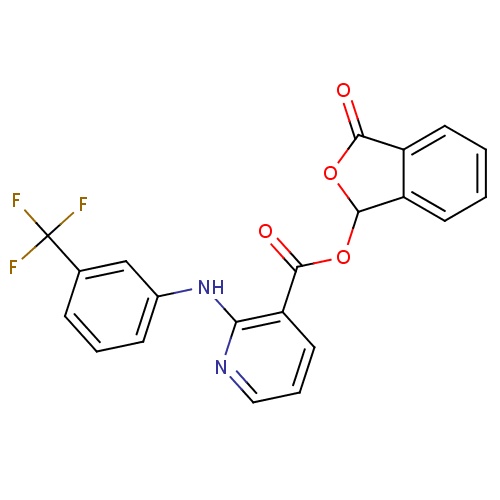
|
; | For the treatment of cystic fibrosis. |
205 ,
21
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2gh6_ligand_frag_9.mol2 | 2gh6 | 1 | -5.41 | C(F)(F)F | 4 |
| 4m3d_ligand_frag_8.mol2 | 4m3d | 1 | -5.38 | C(F)(F)F | 4 |
| 1ela_ligand_frag_1.mol2 | 1ela | 1 | -5.37 | C(F)(F)F | 4 |
| 1ele_ligand_frag_1.mol2 | 1ele | 1 | -5.37 | C(F)(F)F | 4 |
| 4m3b_ligand_frag_0.mol2 | 4m3b | 1 | -5.37 | FC(F)F | 4 |
| 4m3e_ligand_frag_0.mol2 | 4m3e | 1 | -5.37 | FC(F)F | 4 |
605 ,
61

