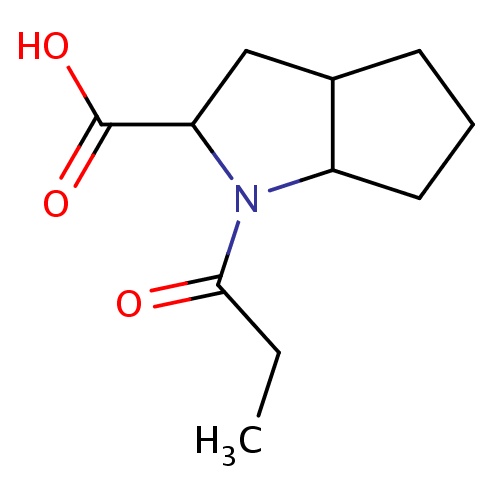
Common name
(2S,3aS,6aS)-1-propanoyl-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid
IUPAC name
(2S,3aS,6aS)-1-propanoyl-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid
SMILES
C(C)C(=O)N1C2C(CC1C(=O)O)CCC2
Common name
(2S,3aS,6aS)-1-propanoyl-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid
IUPAC name
(2S,3aS,6aS)-1-propanoyl-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid
SMILES
C(C)C(=O)N1C2C(CC1C(=O)O)CCC2
INCHI
InChI=1S/C11H17NO3/c1-2-10(13)12-8-5-3-4-7(8)6-9(12)11(14)15/h7-9H,2-6H2,1H3,(H,14,15)/t7-,8-,9-/m0/s1
FORMULA
C11H17NO3

Common name
(2S,3aS,6aS)-1-propanoyl-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid
IUPAC name
(2S,3aS,6aS)-1-propanoyl-3,3a,4,5,6,6a-hexahydro-2H-cyclopenta[b]pyrrole-2-carboxylic acid
Molecular weight
211.258
clogP
0.654
clogS
-0.585
Frequency
0.0003
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
57.61
Number of Rings
2
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00067 | Ramipril |
|
Angiotensin-Converting Enzyme Inhibitors; Antihypertensive Agents; Lipid Modifying Agents; Cardiovascular System; Agents Acting on the Renin-Angiotensin System; ACE Inhibitors, Plain; ACE Inhibitors and Diuretics; ACE Inhibitors and Calcium Channel Blockers; | For the management of mild to severe hypertension. May be used to reduce cardiovascular mortality following myocardial infarction in hemodynamically stable individuals who develop clinical signs of congestive heart failure within a few days following myocardial infarction. To reduce the rate of death, myocardial infarction and stroke in individuals at high risk of cardiovascular events. May be used to slow the progression of renal disease in individuals with hypertension, diabetes mellitus and microalubinuria or overt nephropathy. |
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4avt_ligand.mol2 | 4avt | 0.943396 | -5.96 | N1([C@H](CCC1)C(=O)O)C(=O)CCCCC(=O)N1[C@H](CCC1)C(=O)O | 25 |
| 4tw6_ligand_2_54.mol2 | 4tw6 | 0.888889 | -7.33 | C(=O)(N1CCCC[C@H]1C(=O)O)CC | 13 |
| 4rn6_ligand_2_11.mol2 | 4rn6 | 0.888889 | -6.98 | CCC(=O)N1[C@H](C[C@@H](CC1)C)C(=O)O | 14 |
| 4kz6_ligand_1_0.mol2 | 4kz6 | 0.888889 | -6.86 | CC(=O)N1[C@H](C)CCC[C@@H]1C(=O)O | 13 |
| 4hfp_ligand_2_11.mol2 | 4hfp | 0.888889 | -6.85 | C(C(=O)N1[C@H](C[C@@H](CC1)C)C(=O)O)C | 14 |
| 1dwc_ligand_2_11.mol2 | 1dwc | 0.888889 | -6.81 | CCC(=O)N1[C@H](C[C@@H](CC1)C)C(=O)O | 14 |
| 4kz6_ligand_frag_0.mol2 | 4kz6 | 0.888889 | -6.60 | C[C@@H]1CCC[C@@H](N1C=O)C(=O)O | 12 |
| 4ayy_ligand_2_15.mol2 | 4ayy | 0.888889 | -6.51 | CCC(=O)N1[C@H](C[C@@H](CC1)C)C(=O)O | 14 |
| 1k1i_ligand_2_5.mol2 | 1k1i | 0.888889 | -6.12 | C(C(=O)N1CCCC[C@@H]1C(=O)O)C | 13 |

