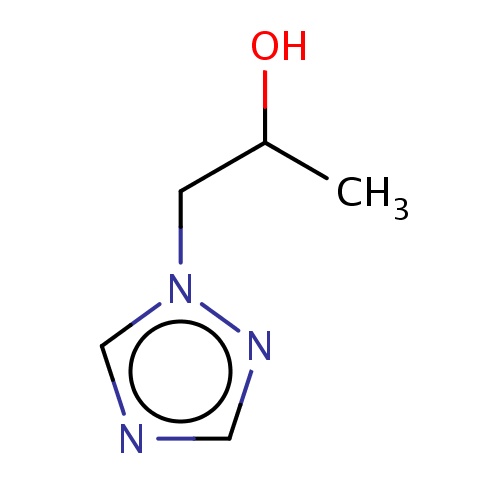
Common name
(2S)-1-(1,2,4-triazol-1-yl)propan-2-ol
IUPAC name
(2S)-1-(1,2,4-triazol-1-yl)propan-2-ol
SMILES
n1(ncnc1)CC(O)C
Common name
(2S)-1-(1,2,4-triazol-1-yl)propan-2-ol
IUPAC name
(2S)-1-(1,2,4-triazol-1-yl)propan-2-ol
SMILES
n1(ncnc1)CC(O)C
INCHI
InChI=1S/C5H9N3O/c1-5(9)2-8-4-6-3-7-8/h3-5,9H,2H2,1H3/t5-/m0/s1
FORMULA
C5H9N3O

Common name
(2S)-1-(1,2,4-triazol-1-yl)propan-2-ol
IUPAC name
(2S)-1-(1,2,4-triazol-1-yl)propan-2-ol
Molecular weight
128.152
clogP
-1.425
clogS
0.036
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
52.79
Number of Rings
1
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00085 | Fluconazole |
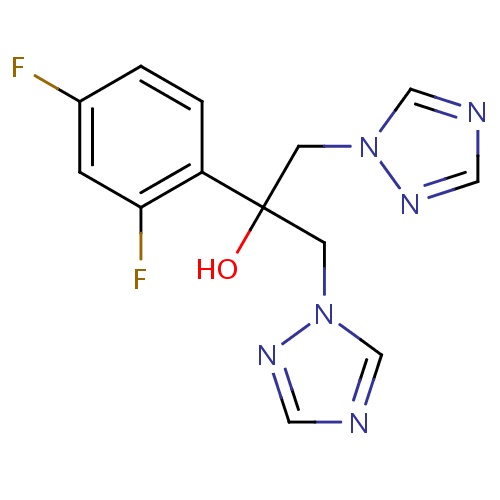
|
Antifungal Agents; 14-alpha Demethylase Inhibitors; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Dermatologicals; Imidazole and Triazole Derivatives; Antifungals for Topical Use; Antifungals for Dermatological Use; Triazole Derivatives; Antimycotics for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of fungal infections. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2c1p_ligand_3_3.mol2 | 2c1p | 1 | -5.92 | c1[nH][n+](cn1)C[C@@H](C)O | 9 |
| 2c1p_ligand_2_1.mol2 | 2c1p | 0.882353 | -5.53 | c1[nH][n+](cn1)CCO | 8 |
| 2c1p_ligand_1_2.mol2 | 2c1p | 0.558824 | -5.48 | c1[nH][n+](cn1)C | 6 |
| 5afv_ligand_1_4.mol2 | 5afv | 0.558824 | -5.48 | C[n+]1c[nH]cn1 | 6 |
| 2c5y_ligand_1_0.mol2 | 2c5y | 0.558824 | -5.44 | C[n+]1c[nH]cn1 | 6 |
| 2c5x_ligand_1_0.mol2 | 2c5x | 0.558824 | -5.37 | c1[nH]cn[n+]1C | 6 |
| 2wd3_ligand_1_2.mol2 | 2wd3 | 0.558824 | -5.22 | [nH]1cn[n+](c1)C | 6 |
| 4pjt_ligand_frag_2.mol2 | 4pjt | 0.558824 | -5.18 | c1[nH]cn[n+]1C | 6 |
101 ,
11

