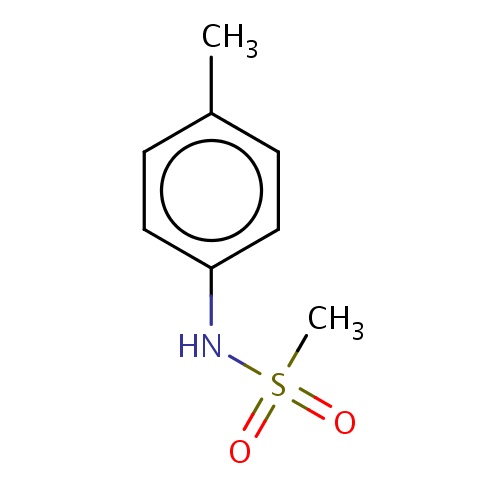
Common name
N-(p-tolyl)methanesulfonamide
IUPAC name
N-(p-tolyl)methanesulfonamide
SMILES
Cc1ccc(cc1)NS(=O)(=O)C
Common name
N-(p-tolyl)methanesulfonamide
IUPAC name
N-(p-tolyl)methanesulfonamide
SMILES
Cc1ccc(cc1)NS(=O)(=O)C
INCHI
InChI=1S/C8H11NO2S/c1-7-3-5-8(6-4-7)9-12(2,10)11/h3-6,9H,1-2H3
FORMULA
C8H11NO2S

Common name
N-(p-tolyl)methanesulfonamide
IUPAC name
N-(p-tolyl)methanesulfonamide
Molecular weight
185.243
clogP
0.795
clogS
-2.544
Frequency
0.0003
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
46.17
Number of Rings
1
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00092 | Dofetilide |
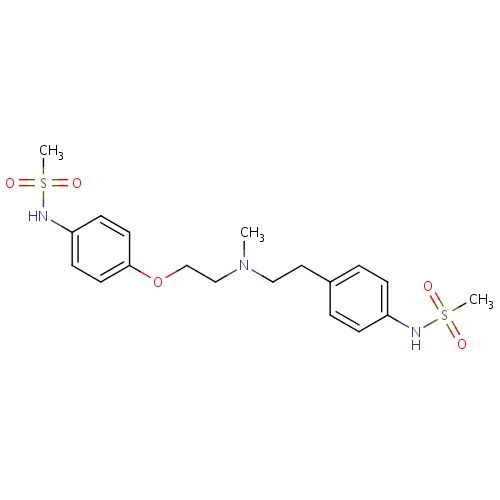
|
Anti-Arrhythmia Agents; Potassium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class III; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; CYP3A4 Inhibitors; | For the maintenance of normal sinus rhythm (delay in time to recurrence of atrial fibrillation/atrial flutter [AF/AFl]) in patients with atrial fibrillation/atrial flutter of greater than one week duration who have been converted to normal sinus rhythm. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4mk8_ligand_2_20.mol2 | 4mk8 | 1 | -6.63 | Cc1ccc(cc1)NS(=O)(=O)C | 12 |
| 2o5k_ligand_2_14.mol2 | 2o5k | 1 | -6.06 | c1(ccc(cc1)NS(=O)(=O)C)C | 12 |
| 3exo_ligand_1_1.mol2 | 3exo | 0.897436 | -6.94 | N(S(=O)(=O)C)c1c(cccc1)C | 12 |
| 1w0y_ligand_2_15.mol2 | 1w0y | 0.897436 | -5.90 | c1(c(cccc1)NS(=O)(=O)C)C | 12 |
| 5bml_ligand_2_0.mol2 | 5bml | 0.875 | -6.57 | Cc1cccc(NS(=O)(=O)C)c1 | 12 |
| 4mk8_ligand_3_34.mol2 | 4mk8 | 0.857143 | -6.75 | CCc1ccc(cc1)NS(=O)(=O)C | 13 |
| 2o5k_ligand_3_19.mol2 | 2o5k | 0.857143 | -6.23 | CCc1ccc(cc1)NS(=O)(=O)C | 13 |
| 4lsj_ligand_1_2.mol2 | 4lsj | 0.805556 | -6.96 | N(S(=O)(=O)C)c1ccccc1 | 11 |
| 4mk8_ligand_1_6.mol2 | 4mk8 | 0.805556 | -6.47 | c1ccc(cc1)NS(=O)(=O)C | 11 |
| 4hxs_ligand_2_3.mol2 | 4hxs | 0.805556 | -6.42 | c1(ccccc1)NS(=O)(=O)C | 11 |
101 ,
11

