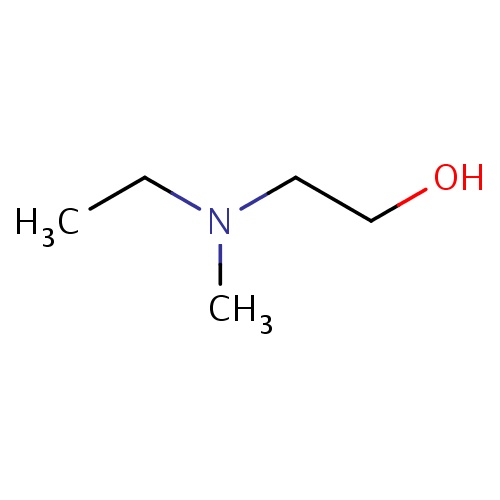
Common name
2-[ethyl(methyl)amino]ethanol
IUPAC name
2-[ethyl(methyl)amino]ethanol
SMILES
C(C)N(CCO)C
Common name
2-[ethyl(methyl)amino]ethanol
IUPAC name
2-[ethyl(methyl)amino]ethanol
SMILES
C(C)N(CCO)C
INCHI
InChI=1S/C5H13NO/c1-3-6(2)4-5-7/h7H,3-5H2,1-2H3
FORMULA
C5H13NO

Common name
2-[ethyl(methyl)amino]ethanol
IUPAC name
2-[ethyl(methyl)amino]ethanol
Molecular weight
103.163
clogP
-0.246
clogS
-0.556
Frequency
0.0010
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
23.47
Number of Rings
0
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
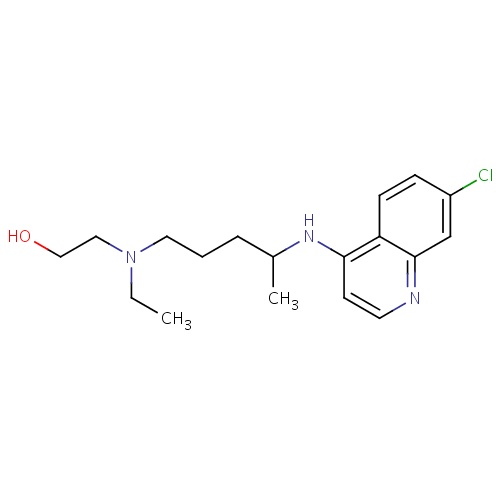
| FDBD00092 | Dofetilide |
|
Anti-Arrhythmia Agents; Potassium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class III; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; CYP3A4 Inhibitors; | For the maintenance of normal sinus rhythm (delay in time to recurrence of atrial fibrillation/atrial flutter [AF/AFl]) in patients with atrial fibrillation/atrial flutter of greater than one week duration who have been converted to normal sinus rhythm. |
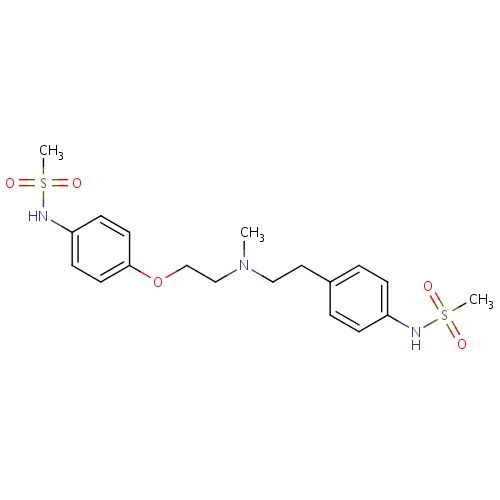
| FDBD01261 | Hydroxychloroquine |
|
Antirheumatic Agents; Enzyme Inhibitors; Antimalarials; Antiprotozoal Agents; Antiparasitic Products, Insecticides and Repellents; Aminoquinolines; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For the suppressive treatment and treatment of acute attacks of malaria due to . |
| FDBD01448 | Lumefantrine |
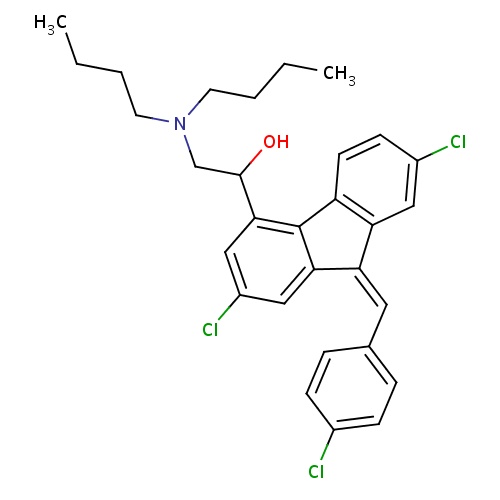
|
Antimalarials; Antiprotozoal Agents; Antiparasitic Products, Insecticides and Repellents; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Lumefantrine and artemether combination therapy is indicated for the treatment of acute uncomplicated malaria caused by . |
3 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3c2u_ligand_3_37.mol2 | 3c2u | 1 | -5.98 | CC[NH2+]CCO | 6 |
| 3c2u_ligand_3_36.mol2 | 3c2u | 1 | -5.89 | CC[NH2+]CCO | 6 |
| 2xfk_ligand_2_68.mol2 | 2xfk | 1 | -5.83 | C(C)[NH2+]CCO | 6 |
| 2viy_ligand_2_49.mol2 | 2viy | 1 | -5.82 | C([NH2+]CC)CO | 6 |
| 2xfi_ligand_2_10.mol2 | 2xfi | 1 | -5.82 | C([NH2+]CC)CO | 6 |
| 2viz_ligand_2_63.mol2 | 2viz | 1 | -5.81 | C(O)C[NH2+]CC | 6 |
| 2vj6_ligand_2_56.mol2 | 2vj6 | 1 | -5.80 | C(O)C[NH2+]CC | 6 |
| 2xfj_ligand_2_56.mol2 | 2xfj | 1 | -5.79 | C([NH2+]CC)CO | 6 |
| 4qkx_ligand_3_28.mol2 | 4qkx | 1 | -5.75 | CC[NH2+]CCO | 6 |
| 3c2u_ligand_3_38.mol2 | 3c2u | 1 | -5.70 | CC[NH2+]CCO | 6 |
195 ,
20

