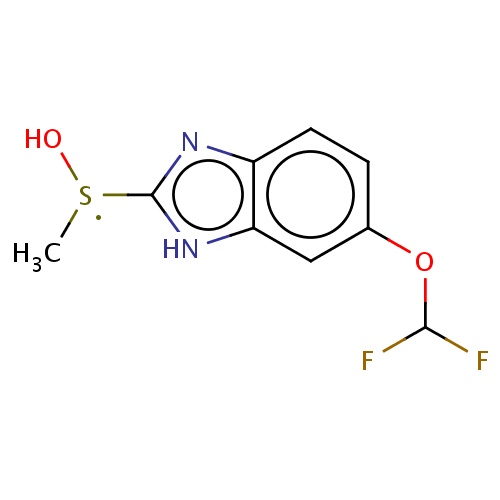
Common name
6-(difluoromethoxy)-2-[hydroxy(methyl)-λ3-sulfanyl]-1H-benzimidazole
IUPAC name
6-(difluoromethoxy)-2-[hydroxy(methyl)-λ3-sulfanyl]-1H-benzimidazole
SMILES
[S](O)(c1[nH]c2c(n1)ccc(c2)OC(F)F)C
Common name
6-(difluoromethoxy)-2-[hydroxy(methyl)-λ3-sulfanyl]-1H-benzimidazole
IUPAC name
6-(difluoromethoxy)-2-[hydroxy(methyl)-λ3-sulfanyl]-1H-benzimidazole
SMILES
[S](O)(c1[nH]c2c(n1)ccc(c2)OC(F)F)C
INCHI
InChI=1S/C9H9F2N2O2S/c1-16(14)9-12-6-3-2-5(15-8(10)11)4-7(6)13-9/h2-4,8,14H,1H3,(H,12,13)
FORMULA
C9H9F2N2O2S

Common name
6-(difluoromethoxy)-2-[hydroxy(methyl)-λ3-sulfanyl]-1H-benzimidazole
IUPAC name
6-(difluoromethoxy)-2-[hydroxy(methyl)-λ3-sulfanyl]-1H-benzimidazole
Molecular weight
247.242
clogP
0.876
clogS
-2.737
Frequency
0.0003
HBond Acceptor
3
HBond Donor
2
Total PolarSurface Area
58.14
Number of Rings
2
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00101 | Pantoprazole |
|
Anti-Ulcer Agents; Proton Pump Inhibitors; Alimentary Tract and Metabolism; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | Short-term (up to 16 weeks) treatment of erosive esophagitis. |
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2i40_ligand_1_0.mol2 | 2i40 | 0.64 | -6.41 | c1(cc2[nH+]c[nH]c2cc1)OC | 11 |
| 2i40_ligand_1_5.mol2 | 2i40 | 0.64 | -6.37 | O(C)c1ccc2[nH+]c[nH]c2c1 | 11 |
| 2i40_ligand_2_4.mol2 | 2i40 | 0.615385 | -6.32 | O(C)c1c(cc2[nH+]c[nH]c2c1)OC | 13 |
| 2qu5_ligand_2_5.mol2 | 2qu5 | 0.578313 | -7.92 | c1cnccc1Oc1cc2c(cc1)[nH+]c[nH]2 | 16 |
| 2xnm_ligand_2_18.mol2 | 2xnm | 0.536082 | -6.83 | [nH+]1c[nH]c2ccc(cc12)O[C@@H]1CC[N@@H+](CC1)C | 17 |
| 2xnp_ligand_2_22.mol2 | 2xnp | 0.536082 | -6.83 | O([C@@H]1CC[N@@H+](CC1)C)c1ccc2[nH]c[nH+]c2c1 | 17 |

