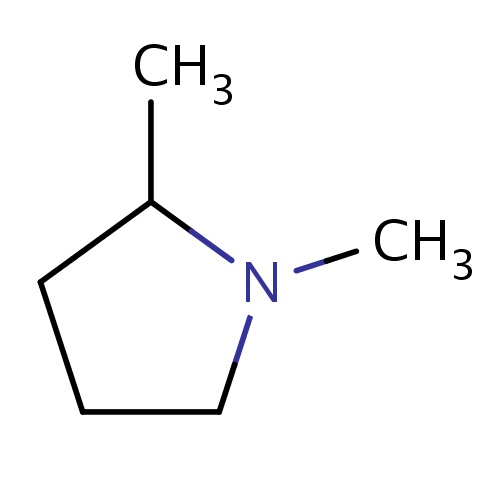
Common name
(2S)-1,2-dimethylpyrrolidine
IUPAC name
(2S)-1,2-dimethylpyrrolidine
SMILES
CC1N(CCC1)C
Common name
(2S)-1,2-dimethylpyrrolidine
IUPAC name
(2S)-1,2-dimethylpyrrolidine
SMILES
CC1N(CCC1)C
INCHI
InChI=1S/C6H13N/c1-6-4-3-5-7(6)2/h6H,3-5H2,1-2H3/t6-/m0/s1
FORMULA
C6H13N

Common name
(2S)-1,2-dimethylpyrrolidine
IUPAC name
(2S)-1,2-dimethylpyrrolidine
Molecular weight
99.174
clogP
1.166
clogS
-0.788
Frequency
0.0010
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
3.24
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
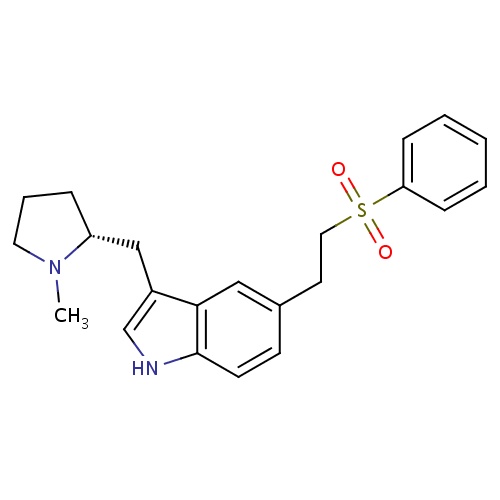
| FDBD00104 | Eletriptan |
|
Analgesics; Serotonin Antagonists; Serotonin Receptor Agonists; Anti-migraine Agents; Nervous System; Selective Serotonin (5Ht1) Agonists; Antimigraine Preparations; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the acute treatment of migraine with or without aura in adults. |
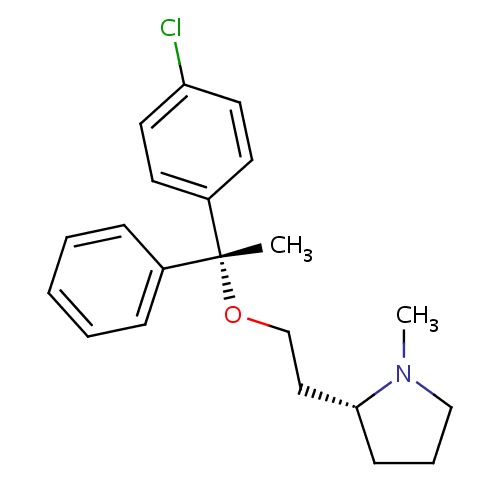
| FDBD00170 | Clemastine |
|
Anti-Allergic Agents; Antipruritics; Histamine H1 Antagonists; Respiratory System; Dermatologicals; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Antihistamines for Topical Use; Aminoalkyl Ethers; Antihistamines for Systemic Use; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the relief of symptoms associated with allergic rhinitis such as sneezing, rhinorrhea, pruritus and acrimation. Also for the management of mild, uncomplicated allergic skin manifestations of urticaria and angioedema. Used as self-medication for temporary relief of symptoms associated with the common cold. |
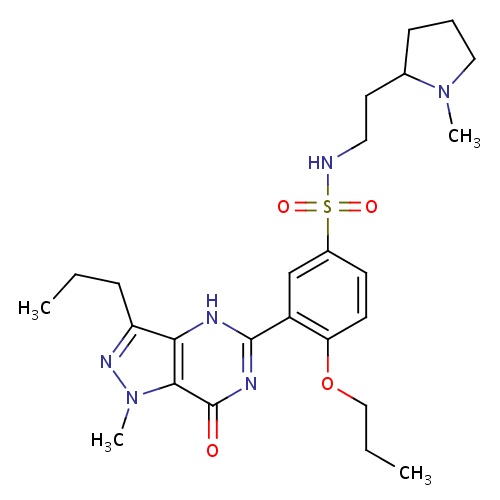
| FDBD01400 | Udenafil |
|
Genito Urinary System and Sex Hormones; Drugs Used in Erectile Dysfunction; Urological Agents; CYP3A4 Inhibitors; | Investigated for use/treatment in erectile dysfunction and hypertension. |
3 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3fh7_ligand_2_22.mol2 | 3fh7 | 1 | -6.47 | C[C@@H]1CCC[N@@H+]1C | 7 |
| 1o0d_ligand_1_1.mol2 | 1o0d | 1 | -6.19 | C1[N@H+]([C@H](CC1)C)C | 7 |
| 2qu6_ligand_1_7.mol2 | 2qu6 | 1 | -5.93 | [N@H+]1(CCC[C@H]1C)C | 7 |
| 4mds_ligand_1_4.mol2 | 4mds | 1 | -5.78 | C[C@H]1CCC[N@@H+]1C | 7 |
| 1szm_ligand_1_0.mol2 | 1szm | 1 | -5.67 | C[C@H]1[N@@H+](C)CCC1 | 7 |
| 3fh7_ligand_3_52.mol2 | 3fh7 | 0.944444 | -6.78 | C[C@@H]1CCC[N@@H+]1CC | 8 |
| 2fxr_ligand_2_45.mol2 | 2fxr | 0.944444 | -6.59 | [C@H]1(CC[NH2+]C1)CC | 7 |
134 ,
14

