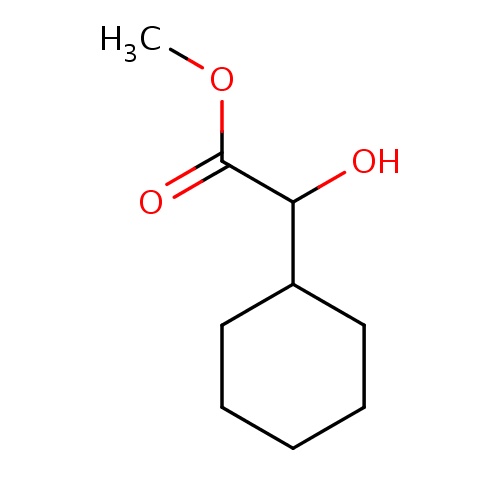
Common name
methyl (2R)-2-cyclohexyl-2-hydroxy-acetate
IUPAC name
methyl (2R)-2-cyclohexyl-2-hydroxy-acetate
SMILES
C1(CCCCC1)C(O)C(=O)OC
Common name
methyl (2R)-2-cyclohexyl-2-hydroxy-acetate
IUPAC name
methyl (2R)-2-cyclohexyl-2-hydroxy-acetate
SMILES
C1(CCCCC1)C(O)C(=O)OC
INCHI
InChI=1S/C9H16O3/c1-12-9(11)8(10)7-5-3-2-4-6-7/h7-8,10H,2-6H2,1H3/t8-/m1/s1
FORMULA
C9H16O3

Common name
methyl (2R)-2-cyclohexyl-2-hydroxy-acetate
IUPAC name
methyl (2R)-2-cyclohexyl-2-hydroxy-acetate
Molecular weight
172.222
clogP
1.347
clogS
-0.862
Frequency
0.0010
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
46.53
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
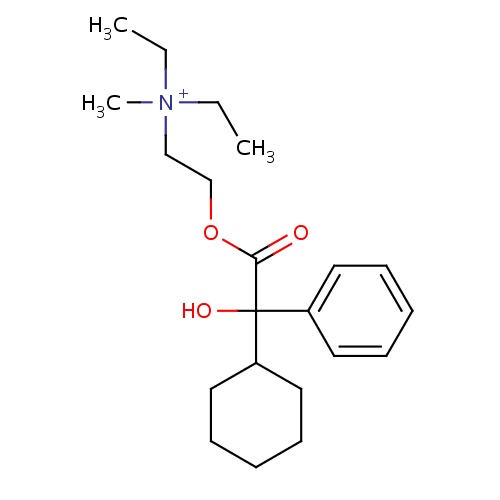
| FDBD00107 | Oxyphenonium |
|
Muscarinic Antagonists; Parasympatholytics; Mydriatics; Alimentary Tract and Metabolism; Drugs for Functional Gastrointestinal Disorders; Synthetic Anticholinergics, Quaternary Ammonium Compounds; | For the treatment of visceral spasms. |
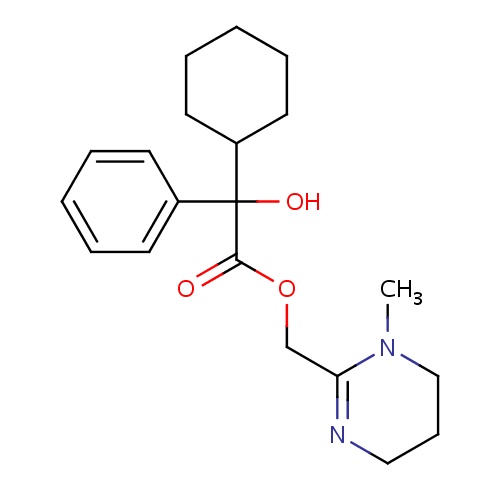
| FDBD00264 | Oxyphencyclimine |
|
Cholinergic Antagonists; Antispasmodics; Alimentary Tract and Metabolism; Drugs for Functional Gastrointestinal Disorders; Synthetic Anticholinergics, Esters With Tertiary Amino Group; | For the treatment of peptic ulcer disease and the relief of smooth muscle spasms in gastrointestinal disorders. |
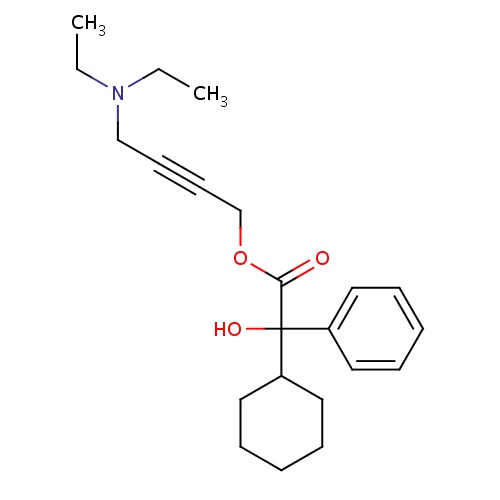
| FDBD00913 | Oxybutynin |
|
Muscarinic Antagonists; Parasympatholytics; Cholinergic Antagonists; Antispasmodics; Muscle Relaxants, Genitourinary; Genito Urinary System and Sex Hormones; Drugs for Urinary Frequency and Incontinence; Urological Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of overactive bladder. |
3 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4foc_ligand_1_0.mol2 | 4foc | 0.896552 | -6.04 | COC(=O)C1CCCCC1 | 10 |
| 4c16_ligand_3_0.mol2 | 4c16 | 0.758621 | -5.49 | O[C@H](C(=O)O)CC1CCCCC1 | 12 |
| 5d1r_ligand.mol2 | 5d1r | 0.757576 | -8.07 | C(CCCCCCCCC)CC(=O)OC(C)C | 18 |
| 4ks3_ligand_2_15.mol2 | 4ks3 | 0.757576 | -6.06 | CO[C@H]1CCC[C@@H](C1)C(=O)O | 11 |
| 2ay2_ligand.mol2 | 2ay2 | 0.689655 | -7.50 | C1(CCCCC1)CCC(=O)O | 12 |
| 1ceb_ligand_frag_0.mol2 | 1ceb | 0.689655 | -6.89 | C1(CCCCC1)C(=O)O | 9 |
| 2xk3_ligand_frag_3.mol2 | 2xk3 | 0.689655 | -6.79 | C1CCC(CC1)C(=O)O | 9 |
| 4p1u_ligand_frag_3.mol2 | 4p1u | 0.689655 | -6.73 | C1[C@H]([C@H]2CC[C@@H]1CC2)C(=O)O | 11 |
106 ,
11

