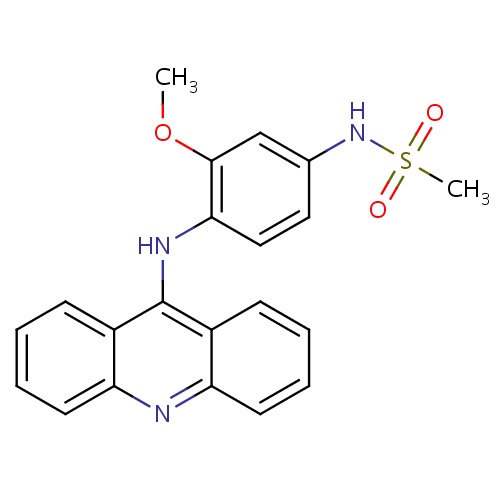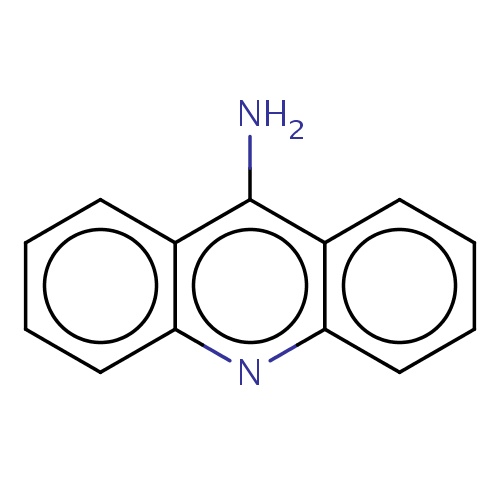
Common name
acridin-9-amine
IUPAC name
acridin-9-amine
SMILES
n1c2c(c(c3c1cccc3)N)cccc2
Common name
acridin-9-amine
IUPAC name
acridin-9-amine
SMILES
n1c2c(c(c3c1cccc3)N)cccc2
INCHI
InChI=1S/C13H10N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1-8H,(H2,14,15)
FORMULA
C13H10N2

Common name
acridin-9-amine
IUPAC name
acridin-9-amine
Molecular weight
194.232
clogP
2.771
clogS
-3.810
Frequency
0.0003
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
38.91
Number of Rings
3
Rotatable Bond
0
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2xui_ligand_1_11.mol2 | 2xui | 1 | -8.04 | c1(c2ccccc2nc2ccccc12)N | 15 |
| 2xup_ligand_1_11.mol2 | 2xup | 1 | -8.03 | Nc1c2ccccc2nc2ccccc12 | 15 |
| 4bds_ligand.mol2 | 4bds | 1 | -7.91 | c1cccc2nc3ccccc3c(c12)N | 16 |
| 2xuf_ligand_1_11.mol2 | 2xuf | 1 | -7.81 | c1(c2ccccc2nc2ccccc12)N | 15 |
| 3frg_ligand_1_2.mol2 | 3frg | 1 | -6.99 | Nc1ccnc2ccccc12 | 11 |
| 2zaz_ligand_1_1.mol2 | 2zaz | 1 | -6.67 | c1cc2c(cc1)c(ccn2)N | 11 |
| 4qt0_ligand_1_1.mol2 | 4qt0 | 1 | -6.57 | Nc1ccnc2c1cccc2 | 11 |
| 4qsm_ligand_1_1.mol2 | 4qsm | 1 | -6.51 | c1c(c2c(nc1)cccc2)N | 11 |
| 1qvt_ligand.mol2 | 1qvt | 0.964286 | -7.97 | c1cc(cc2nc3cc(ccc3cc12)N)N | 17 |
| 1qvu_ligand.mol2 | 1qvu | 0.964286 | -7.89 | c1cc(cc2nc3cc(ccc3cc12)N)N | 17 |
171 ,
18