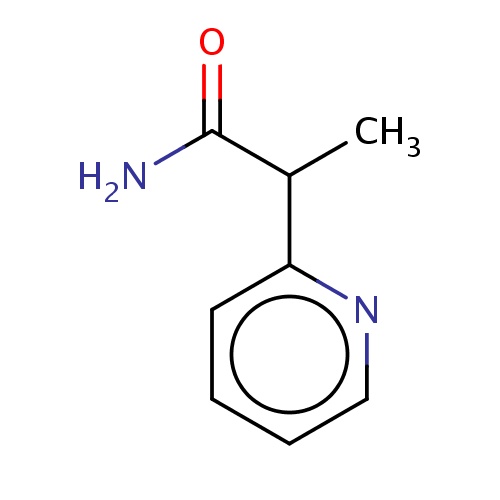
Common name
(2S)-2-(2-pyridyl)propanamide
IUPAC name
(2S)-2-(2-pyridyl)propanamide
SMILES
O=C(N)C(c1ncccc1)C
Common name
(2S)-2-(2-pyridyl)propanamide
IUPAC name
(2S)-2-(2-pyridyl)propanamide
SMILES
O=C(N)C(c1ncccc1)C
INCHI
InChI=1S/C8H10N2O/c1-6(8(9)11)7-4-2-3-5-10-7/h2-6H,1H3,(H2,9,11)/t6-/m0/s1
FORMULA
C8H10N2O

Common name
(2S)-2-(2-pyridyl)propanamide
IUPAC name
(2S)-2-(2-pyridyl)propanamide
Molecular weight
150.178
clogP
0.953
clogS
-1.426
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
55.98
Number of Rings
1
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00167 | Disopyramide |
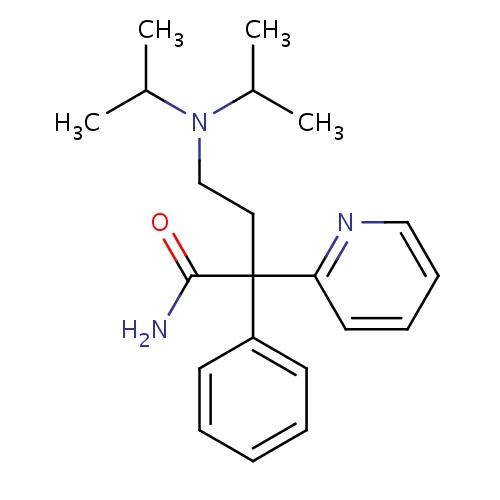
|
Anti-Arrhythmia Agents; Voltage-Gated Sodium Channel Blockers; Cardiovascular System; Antiarrhythmics, Class I and Iii; Cardiac Therapy; Antiarrythmics, Class I and Iii; Antiarrhythmics, Class Ia; CYP3A4 Inhibitors; | For the treatment of documented ventricular arrhythmias, such as sustained ventricular tachycardia, ventricular pre-excitation and cardiac dysrhythmias. It is a Class Ia antiarrhythmic drug. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2bxt_ligand_3_55.mol2 | 2bxt | 0.818182 | -6.02 | c1c(nccc1)CC[NH3+] | 9 |
| 2bvx_ligand_3_55.mol2 | 2bvx | 0.818182 | -5.87 | c1c(nccc1)CC[NH3+] | 9 |
| 2bxu_ligand_3_0.mol2 | 2bxu | 0.661765 | -6.18 | C(F)(F)(c1ncccc1)C[NH3+] | 11 |
| 1mu6_ligand_3_0.mol2 | 1mu6 | 0.661765 | -6.09 | [NH3+]CC(F)(F)c1ncccc1 | 11 |
| 1mu8_ligand_3_0.mol2 | 1mu8 | 0.661765 | -6.07 | C(F)(F)(c1ncccc1)C[NH3+] | 11 |
| 4jps_ligand_1_0.mol2 | 4jps | 0.654545 | -6.43 | C(C)(C)c1ncccc1 | 9 |
| 4ya8_ligand_1_13.mol2 | 4ya8 | 0.654545 | -6.40 | c1(ncccc1)C(C)C | 9 |
| 3frz_ligand_1_6.mol2 | 3frz | 0.654545 | -6.19 | C(C)c1ncccc1 | 8 |
125 ,
13

