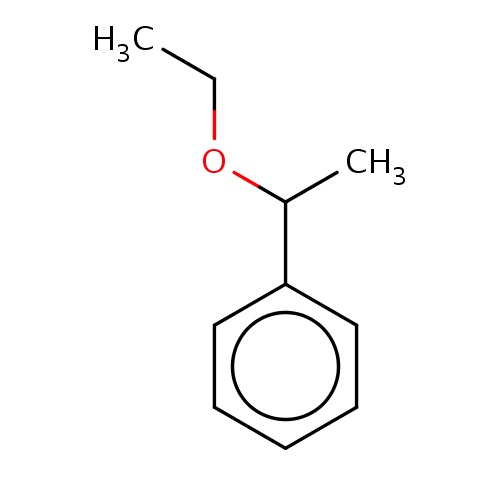
Common name
[(1R)-1-ethoxyethyl]benzene
IUPAC name
[(1R)-1-ethoxyethyl]benzene
SMILES
O(C(C)c1ccccc1)CC
Common name
[(1R)-1-ethoxyethyl]benzene
IUPAC name
[(1R)-1-ethoxyethyl]benzene
SMILES
O(C(C)c1ccccc1)CC
INCHI
InChI=1S/C10H14O/c1-3-11-9(2)10-7-5-4-6-8-10/h4-9H,3H2,1-2H3/t9-/m1/s1
FORMULA
C10H14O

Common name
[(1R)-1-ethoxyethyl]benzene
IUPAC name
[(1R)-1-ethoxyethyl]benzene
Molecular weight
150.218
clogP
2.672
clogS
-2.590
Frequency
0.0007
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
9.23
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
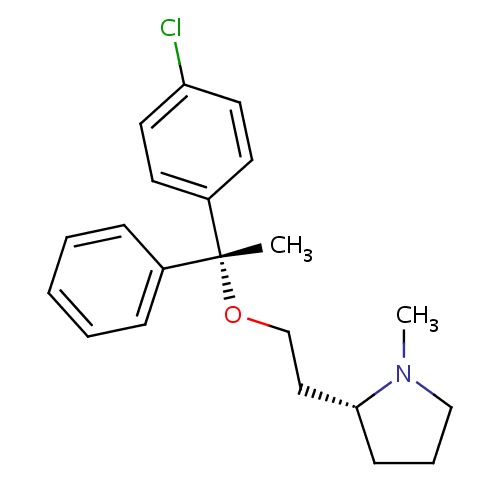
| FDBD00170 | Clemastine |
|
Anti-Allergic Agents; Antipruritics; Histamine H1 Antagonists; Respiratory System; Dermatologicals; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Antihistamines for Topical Use; Aminoalkyl Ethers; Antihistamines for Systemic Use; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the relief of symptoms associated with allergic rhinitis such as sneezing, rhinorrhea, pruritus and acrimation. Also for the management of mild, uncomplicated allergic skin manifestations of urticaria and angioedema. Used as self-medication for temporary relief of symptoms associated with the common cold. |
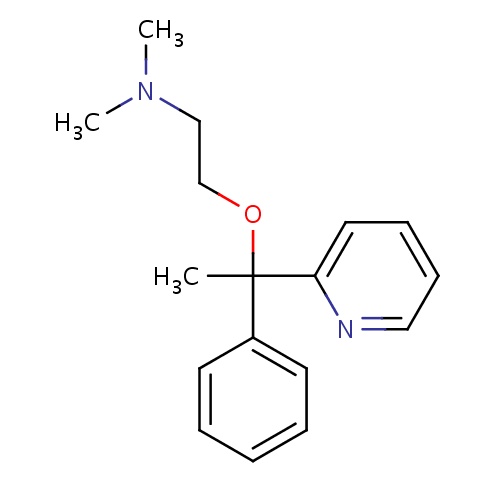
| FDBD00249 | Doxylamine |
|
Histamine H1 Antagonists; Antiemetics; Respiratory System; Aminoalkyl Ethers; Antihistamines for Systemic Use; | Used alone as a short-term sleep aid, in combination with other drugs as a night-time cold and allergy relief drug. Also used in combination with Vitamin B6 (pyridoxine) to prevent morning sickness in pregnant women. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2bkt_ligand_4_172.mol2 | 2bkt | 0.833333 | -7.41 | c1c(cccc1)COCC | 10 |
| 2bks_ligand_4_371.mol2 | 2bks | 0.833333 | -7.09 | CCOCc1ccccc1 | 10 |
| 1d4i_ligand_4_243.mol2 | 1d4i | 0.833333 | -6.44 | C(C)OCc1ccccc1 | 10 |
| 1ms6_ligand_4_198.mol2 | 1ms6 | 0.833333 | -6.36 | c1(ccccc1)COCC | 10 |
| 2y8c_ligand_4_482.mol2 | 2y8c | 0.833333 | -6.08 | CCOCc1ccccc1 | 10 |
| 2xno_ligand_4_295.mol2 | 2xno | 0.833333 | -5.95 | c1(ccccc1)COCC | 10 |
| 1vj9_ligand_4_0.mol2 | 1vj9 | 0.833333 | -5.78 | C(C)OCc1ccccc1 | 10 |
| 2y8c_ligand_4_480.mol2 | 2y8c | 0.833333 | -5.75 | CCOCc1ccccc1 | 10 |
| 2y8c_ligand_4_481.mol2 | 2y8c | 0.833333 | -5.69 | CCOCc1ccccc1 | 10 |
103 ,
11

