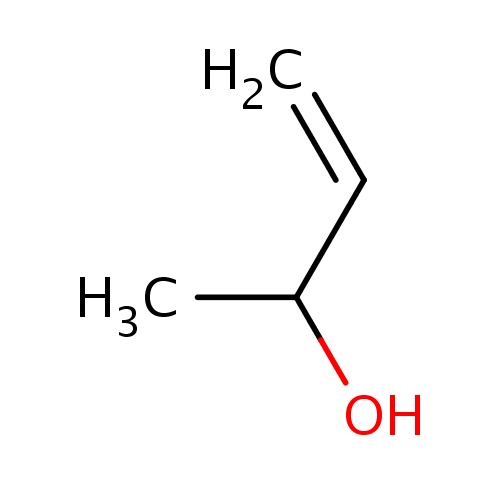
Common name
(2S)-but-3-en-2-ol
IUPAC name
(2S)-but-3-en-2-ol
SMILES
C=CC(O)C
Common name
(2S)-but-3-en-2-ol
IUPAC name
(2S)-but-3-en-2-ol
SMILES
C=CC(O)C
INCHI
InChI=1S/C4H8O/c1-3-4(2)5/h3-5H,1H2,2H3/t4-/m0/s1
FORMULA
C4H8O

Common name
(2S)-but-3-en-2-ol
IUPAC name
(2S)-but-3-en-2-ol
Molecular weight
72.106
clogP
0.257
clogS
-0.047
Frequency
0.0031
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
20.23
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00174 | Travoprost |
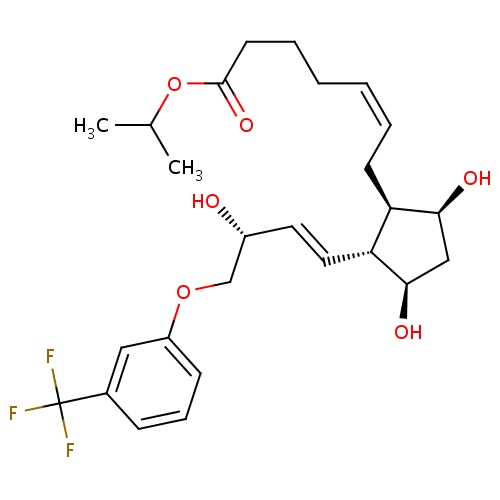
|
Antihypertensive Agents; Prostaglandins, Synthetic; Anti-glaucoma Agents; Ophthalmics; Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | Ophthalmic solution used for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication. |
| FDBD00633 | Alprostadil |
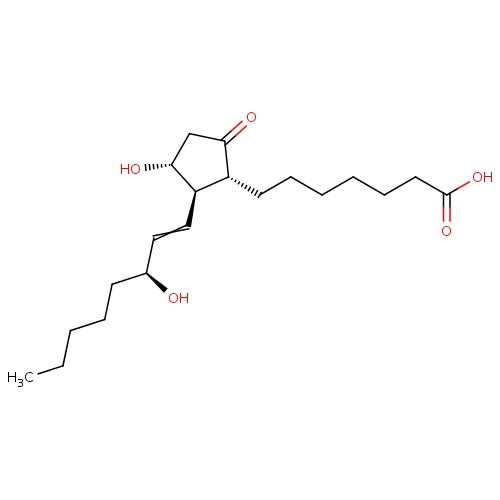
|
Platelet Aggregation Inhibitors; Vasodilator Agents; Prostaglandins; Genito Urinary System and Sex Hormones; Cardiovascular System; Drugs Used in Erectile Dysfunction; Cardiac Therapy; Urological Agents; | For palliative, not definitive, therapy to temporarily maintain the patency of the ductus arteriosus until corrective or palliative surgery can be performed in neonates who have congenital heart defects and who depend upon the patent ductus for survival. Also for the treatment of erectile dysfunction due to neurogenic, vasculogenic, psychogenic, or mixed etiology. |
| FDBD00762 | Bimatoprost |
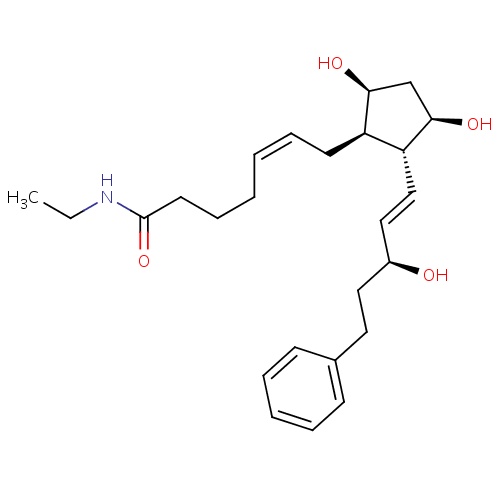
|
Antihypertensive Agents; Anti-glaucoma Agents; Ophthalmologicals; Sensory Organs; Antiglaucoma Preparations and Miotics; Prostaglandin Analogues; | For the reduction of elevated intraocular pressure in patients with open angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication. |
| FDBD00774 | Dinoprostone |
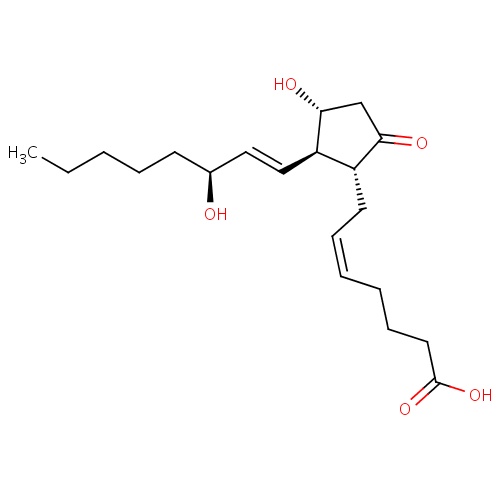
|
For the termination of pregnancy during the second trimester (from the 12th through the 20th gestational week as calculated from the first day of the last normal menstrual period), as well as for evacuation of the uterine contents in the management of missed abortion or intrauterine fetal death up to 28 weeks of gestational age as calculated from the first day of the last normal menstrual period. Also used in the management of nonmetastatic gestational trophoblastic disease (benign hydatidiform mole). Other indications include improving the cervical inducibility (cervical ; | |
| FDBD00945 | Fluvastatin |
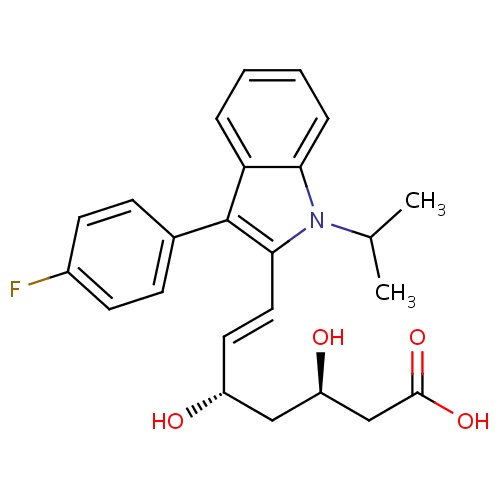
|
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | To be used as an adjunct to dietary therapy to prevent cardiovascular events. May be used as secondary prevention in patients with coronary heart disease (CHD) to reduce the risk of requiring coronary revascularization procedures, for reducing progression of coronary atherosclerosis in hypercholesterolemic patients with CHD, and for the treatment of primary hypercholesterolemia and mixed dyslidipidemia. |
| FDBD00948 | Rosuvastatin |
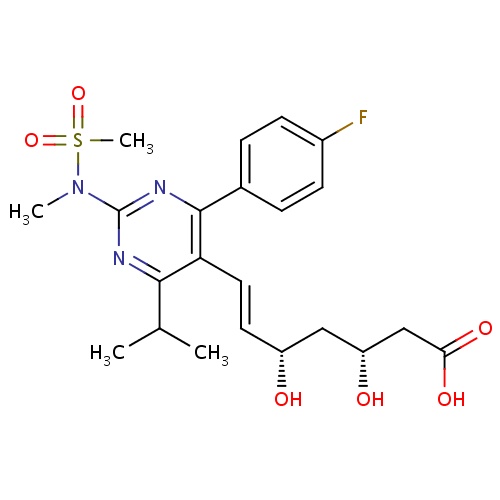
|
Hydroxymethylglutaryl-CoA Reductase Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; | Used as an adjunct to dietary therapy to treat primary hyperlipidemia (heterozygous familial and nonfamilial), mixed dyslipidemia and hypertriglyceridemia. Also indicated for homozygous familial hypercholesterolemia as an adjunct to other lipid-lowering therapies or when other such therapies are not available. Furthermore, it is used to slow the progression of atherosclerosis and for primary prevention of cardiovascular disease. |
| FDBD01084 | Epoprostenol |
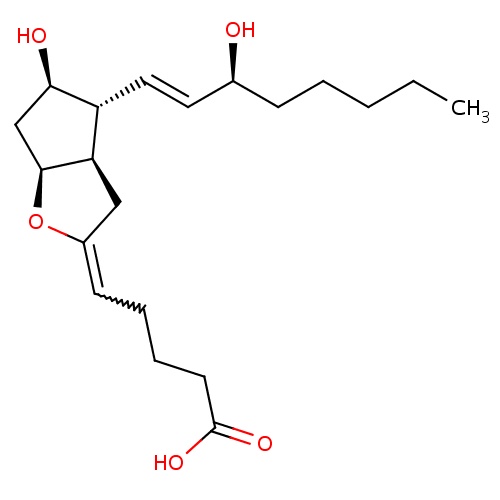
|
Platelet Aggregation Inhibitors; Antihypertensive Agents; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; | For the long-term intravenous treatment of primary pulmonary hypertension and pulmonary hypertension associated with the scleroderma spectrum of disease in NYHA Class III and Class IV patients who do not respond adequately to conventional therapy. |
| FDBD01543 | Pitavastatin |
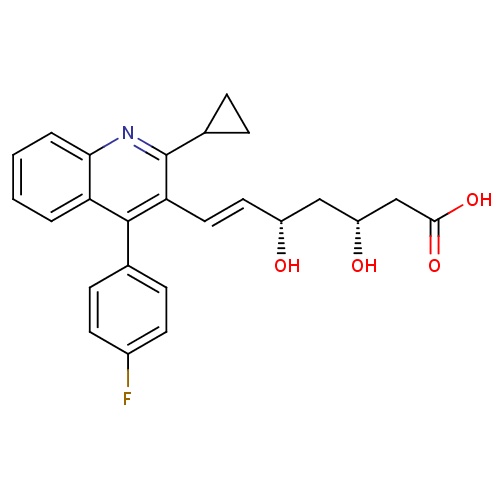
|
Hydroxymethylglutaryl-CoA Reductase Inhibitors; Hypolipidemic Agents; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; | Pitavastatin is used to lower serum levels of total cholesterol, LDL-C, apolipoprotein B, and triglycerides, and raise levels of HDL-C for the treatment of dyslipidemia. |
| FDBD01741 | Limaprost |
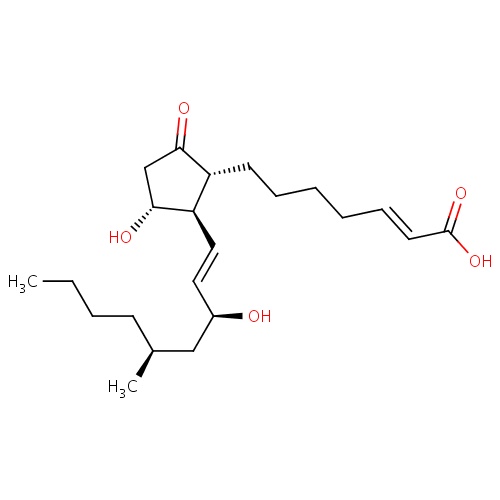
|
; | Limaprost is used for the improvement of various ischemic symptoms such as ulcer, pain and feeling of coldness associated with thromboangiitis obliterans as well as improvement of subjective symptoms (pain and numbness of lower legs) and gait ability associated with acquired lumbar spinal canal stenosis (in patients with bilateral intermittent claudication showing normal SLR test result). |
9 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1o9e_ligand_2_44.mol2 | 1o9e | 1 | -5.53 | C(=C)C(O)(C)C | 6 |
| 2fxu_ligand_1_16.mol2 | 2fxu | 0.8 | -5.56 | [C@H](O)(C)C(=C)C | 6 |
| 3hkw_ligand_2_5.mol2 | 3hkw | 0.777778 | -5.88 | OCC(=C)C | 5 |
| 3hky_ligand_2_5.mol2 | 3hky | 0.777778 | -5.85 | OCC(=C)C | 5 |
| 1o1s_ligand_2_36.mol2 | 1o1s | 0.777778 | -5.62 | C(O)C(=C)C | 5 |
| 4rlw_ligand_1_1.mol2 | 4rlw | 0.75 | -5.63 | C(O)C=C | 4 |
| 3f6e_ligand_1_1.mol2 | 3f6e | 0.75 | -5.60 | C=CCO | 4 |
146 ,
15

