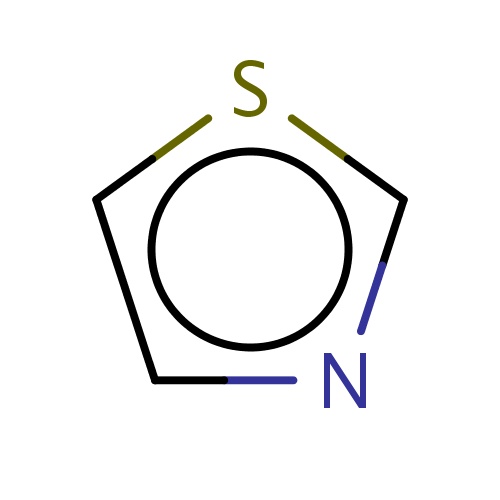
Common name
thiazole
IUPAC name
thiazole
SMILES
s1cncc1
Common name
thiazole
IUPAC name
thiazole
SMILES
s1cncc1
INCHI
InChI=1S/C3H3NS/c1-2-5-3-4-1/h1-3H
FORMULA
C3H3NS

Common name
thiazole
IUPAC name
thiazole
Molecular weight
85.128
clogP
2.480
clogS
-0.732
Frequency
0.0055
HBond Acceptor
2
HBond Donor
0
Total PolarSurface Area
41.13
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00177 | Bleomycin |
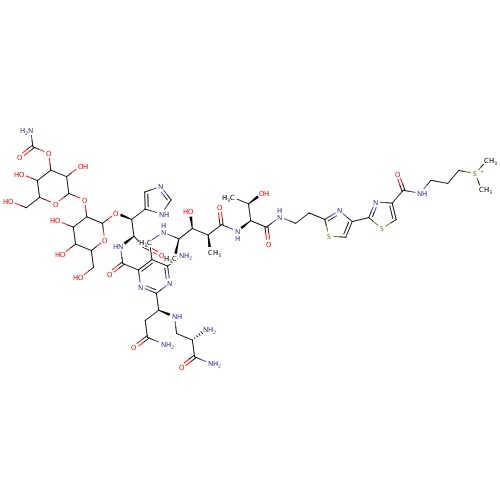
|
Antineoplastic Agents; Immunosuppressive Agents; Antibiotics, Antineoplastic; Antineoplastic and Immunomodulating Agents; Cytotoxic Antibiotics and Related Substances; | For palliative treatment in the management malignant neoplasm (trachea, bronchus, lung), squamous cell carcinoma, and lymphomas. |
| FDBD00376 | Ritonavir |

|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
| FDBD00450 | Nizatidine |
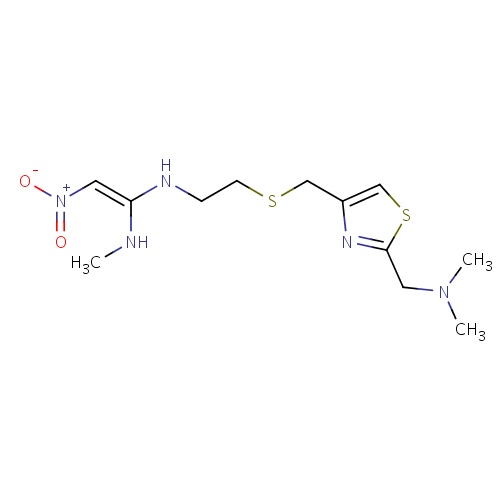
|
Anti-Ulcer Agents; Alimentary Tract and Metabolism; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; H2 Antagonists; | For the treatment of acid-reflux disorders (GERD), peptic ulcer disease, active benign gastric ulcer, and active duodenal ulcer. |
| FDBD00452 | Cinalukast |
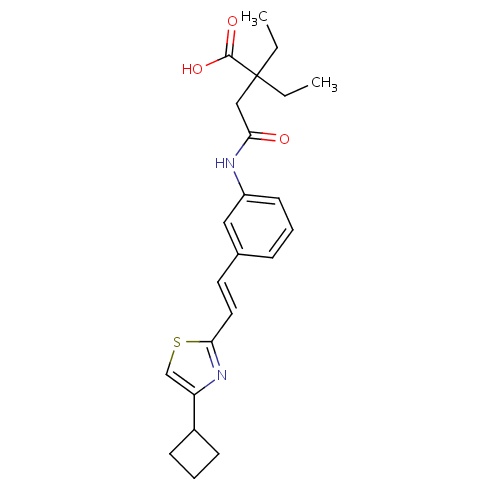
|
Anti-Arrhythmia Agents; | For Protection against second- phase inflamation in exercise-induced bronchoconstriction and Asthma. |
| FDBD00594 | Thiabendazole |
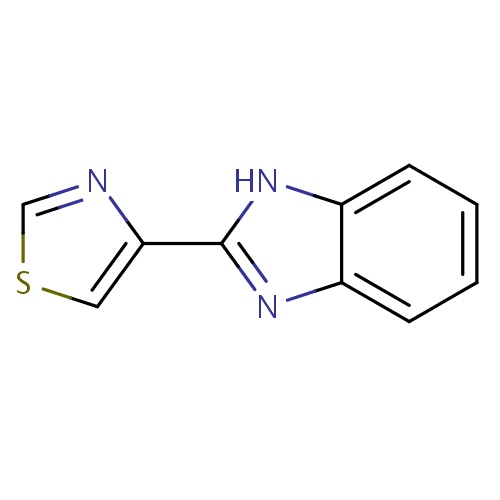
|
Antinematodal Agents; Anthelmintics; Dermatologicals; Antiparasitic Products, Insecticides and Repellents; Imidazole and Triazole Derivatives; Antifungals for Topical Use; Antifungals for Dermatological Use; Benzimidazole Derivatives; Benzimidazoles and Related Substances; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; | For the treatment of strongyloidiasis (threadworm), cutaneous larva migrans (creeping eruption), visceral larva migrans, and trichinosis. |
| FDBD01098 | Dasatinib |

|
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | For the treatment of adults with chronic, accelerated, or myeloid or lymphoid blast phase chronic myeloid leukemia with resistance or intolerance to prior therapy. Also indicated for the treatment of adults with Philadelphia chromosome-positive acute lymphoblastic leukemia with resistance or intolerance to prior therapy. |
| FDBD01375 | Sulfathiazole |
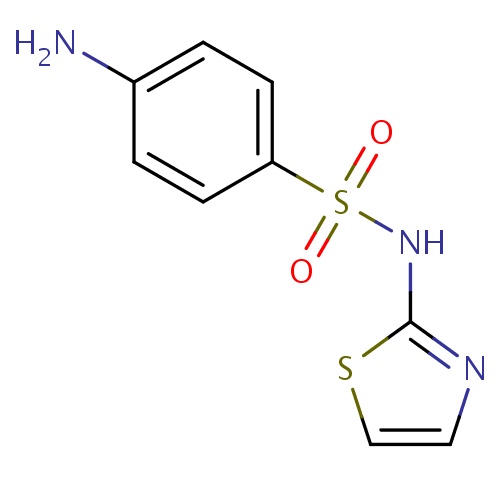
|
Anti-Infective Agents; Sulfonamides; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; Dermatologicals; Short-Acting Sulfonamides; Sulfonamides and Trimethoprim; | Sulfathiazole is effective against a wide range of gram positive and gram negative pathogenic microorganisms. Although no longer used in humans, it is used in cattle. |
| FDBD01406 | Simeprevir |

|
Simeprevir is indicated in patient's with hepatitis C virus (HCV) genotype 1 for the treatment of chronic hepatitis as a combination therapy, which includes peginterferon alfa and ribavirin. | |
| FDBD01420 | Ceftaroline fosamil |
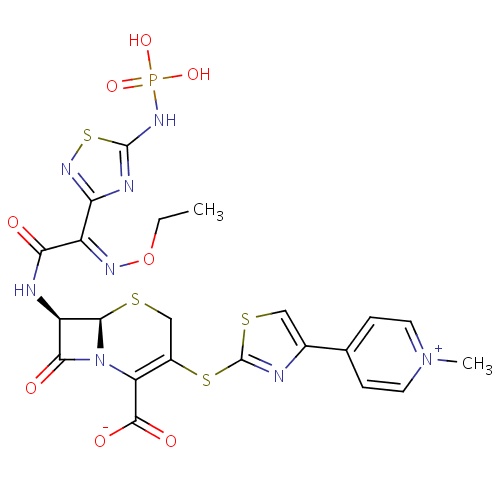
|
Cephalosporins; Antibiotics; Antibacterials for Systemic Use; Antiinfectives for Systemic Use; | Ceftaroline fosamil is indicated for the treatment of patients with the following infections caused by susceptible isolates of the designated microorganisms. |
| FDBD01427 | Isavuconazonium |
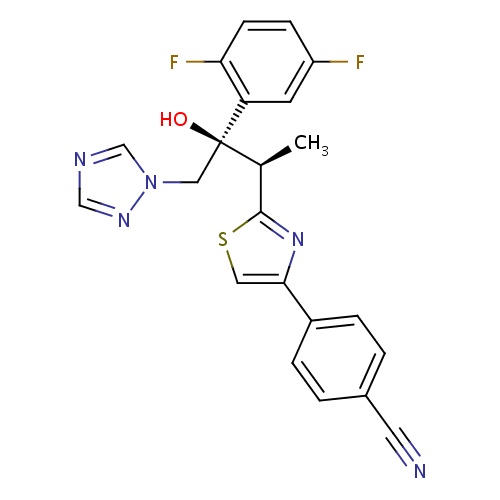
|
CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in the treatment of invasive aspergillosis and invasive mucormycosis. |
16 ,
2
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4g3e_ligand_frag_2.mol2 | 4g3e | 1 | -5.68 | c1cncs1 | 5 |
| 4eqc_ligand_frag_3.mol2 | 4eqc | 1 | -5.50 | c1cncs1 | 5 |
| 4m3e_ligand_frag_5.mol2 | 4m3e | 1 | -5.49 | c1ncsc1 | 5 |
| 4g3f_ligand_frag_1.mol2 | 4g3f | 1 | -5.48 | c1nccs1 | 5 |
| 4g3g_ligand_frag_1.mol2 | 4g3g | 1 | -5.48 | c1nccs1 | 5 |
| 2bts_ligand_frag_1.mol2 | 2bts | 1 | -5.47 | c1cscn1 | 5 |
150 ,
16

