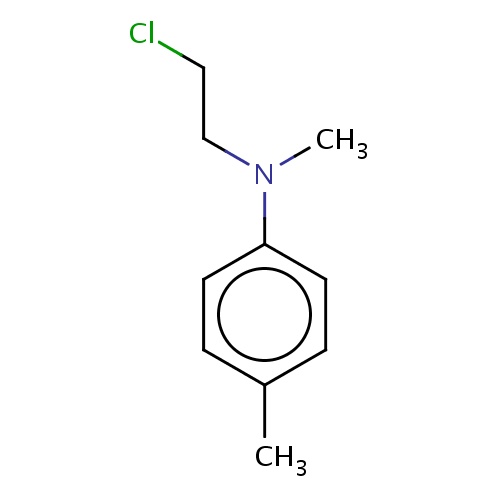
Common name
N-(2-chloroethyl)-N,4-dimethyl-aniline
IUPAC name
N-(2-chloroethyl)-N,4-dimethyl-aniline
SMILES
C(N(C)c1ccc(cc1)C)CCl
Common name
N-(2-chloroethyl)-N,4-dimethyl-aniline
IUPAC name
N-(2-chloroethyl)-N,4-dimethyl-aniline
SMILES
C(N(C)c1ccc(cc1)C)CCl
INCHI
InChI=1S/C10H14ClN/c1-9-3-5-10(6-4-9)12(2)8-7-11/h3-6H,7-8H2,1-2H3
FORMULA
C10H14ClN

Common name
N-(2-chloroethyl)-N,4-dimethyl-aniline
IUPAC name
N-(2-chloroethyl)-N,4-dimethyl-aniline
Molecular weight
183.678
clogP
2.713
clogS
-3.328
Frequency
0.0007
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
3.24
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00178 | Chlorambucil |
|
Antineoplastic Agents; Immunosuppressive Agents; Antineoplastic Agents, Alkylating; Alkylating Agents; Antineoplastic and Immunomodulating Agents; Nitrogen Mustard Analogues; | For treatment of chronic lymphatic (lymphocytic) leukemia, childhood minimal-change nephrotic syndrome, and malignant lymphomas including lymphosarcoma, giant follicular lymphoma, Hodgkin's disease, non-Hodgkin's lymphomas, and Waldenström's Macroglobulinemia. |
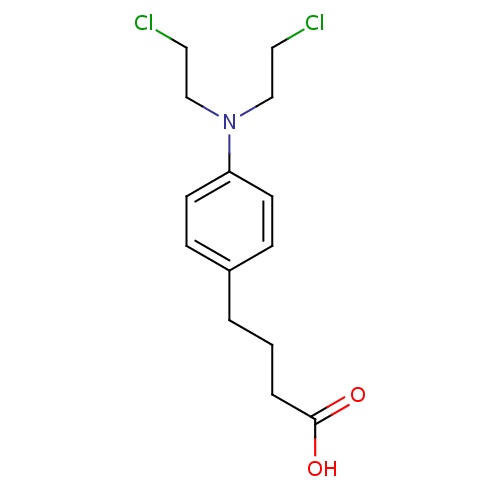
| FDBD00894 | Melphalan |

|
Antineoplastic Agents; Immunosuppressive Agents; Antineoplastic Agents, Alkylating; Alkylating Agents; Myeloablative Agonists; Antineoplastic and Immunomodulating Agents; Nitrogen Mustard Analogues; | For the palliative treatment of multiple myeloma and for the palliation of non-resectable epithelial carcinoma of the ovary. Has also been used alone or as part of various chemotherapeutic regimens as an adjunct to surgery in the treatment of breast cancer, alone or in combination regimens for palliative treatment of locally recurrent or unresectable in-transit metastatic melanoma of the extremities, as well as for the treatment of amyloidosis with prednisone. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4hj2_ligand_5_6167.mol2 | 4hj2 | 1 | -6.73 | C(CCl)N(C)c1ccc(cc1)C | 12 |
| 4hj2_ligand_4_2365.mol2 | 4hj2 | 1 | -6.66 | C(CCl)Nc1ccc(cc1)C | 11 |
| 4hj2_ligand_5_6133.mol2 | 4hj2 | 0.805556 | -6.93 | CN(CC)c1ccc(cc1)C | 11 |
| 4awo_ligand_2_1.mol2 | 4awo | 0.805556 | -6.90 | N(c1ccc(cc1)C)CC | 10 |
| 4hj2_ligand_4_2316.mol2 | 4hj2 | 0.805556 | -6.87 | CCNc1ccc(cc1)C | 10 |
| 4hj2_ligand_4_2345.mol2 | 4hj2 | 0.805556 | -6.48 | C(CCl)N(C)c1ccccc1 | 11 |
| 4hj2_ligand_3_660.mol2 | 4hj2 | 0.805556 | -6.41 | C(CCl)Nc1ccccc1 | 10 |
| 4lnb_ligand_5_456.mol2 | 4lnb | 0.707317 | -5.71 | c1(ccc(cc1)C[NH3+])N(C)CC | 12 |
| 4k3h_ligand_2_5.mol2 | 4k3h | 0.694444 | -7.28 | c1(ccc(cc1)N(C)C)C | 10 |
| 4k3h_ligand_2_8.mol2 | 4k3h | 0.694444 | -7.25 | c1(ccc(cc1)N(C)C)C | 10 |
114 ,
12

