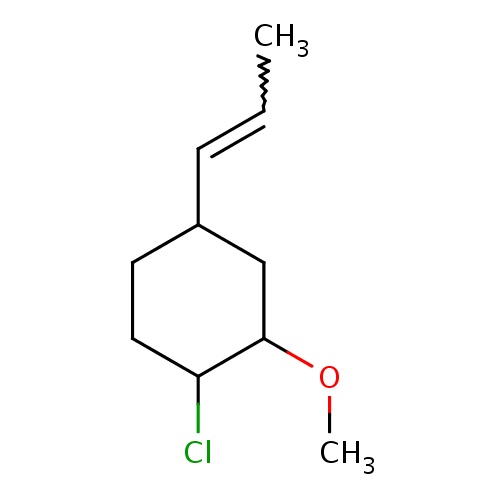
Common name
(1S,2S,4S)-1-chloro-2-methoxy-4-[(E)-prop-1-enyl]cyclohexane
IUPAC name
(1S,2S,4S)-1-chloro-2-methoxy-4-[(E)-prop-1-enyl]cyclohexane
SMILES
C(=CC)C1CC(C(CC1)Cl)OC
Common name
(1S,2S,4S)-1-chloro-2-methoxy-4-[(E)-prop-1-enyl]cyclohexane
IUPAC name
(1S,2S,4S)-1-chloro-2-methoxy-4-[(E)-prop-1-enyl]cyclohexane
SMILES
C(=CC)C1CC(C(CC1)Cl)OC
INCHI
InChI=1S/C10H17ClO/c1-3-4-8-5-6-9(11)10(7-8)12-2/h3-4,8-10H,5-7H2,1-2H3/b4-3+/t8-,9-,10-/m0/s1
FORMULA
C10H17ClO

Common name
(1S,2S,4S)-1-chloro-2-methoxy-4-[(E)-prop-1-enyl]cyclohexane
IUPAC name
(1S,2S,4S)-1-chloro-2-methoxy-4-[(E)-prop-1-enyl]cyclohexane
Molecular weight
188.694
clogP
2.699
clogS
-2.078
Frequency
0.0003
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
9.23
Number of Rings
1
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00222 | Pimecrolimus |
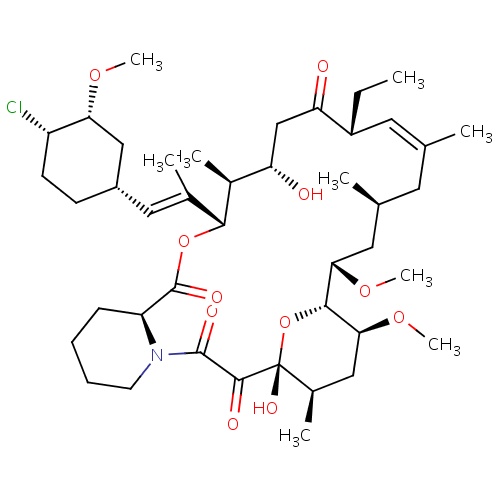
|
Anti-Inflammatory Agents, Non-Steroidal; Dermatologic Agents; Immunosuppressive Agents; Calcineurin Inhibitors; Dermatologicals; Agents for Dermatitis, Excluding Corticosteroids; CYP3A4 Inhibitors; | For treatment of mild to moderate atopic dermatitis. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1qpl_ligand_2_22.mol2 | 1qpl | 0.756098 | -5.75 | [C@@H]1(C[C@H](CCC1)OC)/C=C/C | 11 |
| 3ihz_ligand_2_18.mol2 | 3ihz | 0.704545 | -6.63 | O(C)[C@@H]1C[C@@H](CC[C@H]1O)/C=C\C | 12 |
| 1fkf_ligand_2_12.mol2 | 1fkf | 0.704545 | -6.48 | O(C)[C@@H]1C[C@@H](CC[C@H]1O)/C=C\C | 12 |
| 1a7x_ligand_2_35.mol2 | 1a7x | 0.704545 | -5.90 | O(C)[C@@H]1C[C@@H](CC[C@H]1O)/C=C\C | 12 |
| 4nnr_ligand_2_18.mol2 | 4nnr | 0.704545 | -5.75 | O(C)[C@@H]1C[C@@H](CC[C@H]1O)/C=C\C | 12 |
| 1qpl_ligand_3_52.mol2 | 1qpl | 0.704545 | -5.69 | [C@@H]1(C[C@H]([C@@H](CC1)O)OC)/C=C/C | 12 |
| 1o9e_ligand_2_17.mol2 | 1o9e | 0.568627 | -7.71 | O(C)C[C@@H]1C2=C[C@@]3([C@@H](CC[C@@H]3O)C[C@@H]([C@@H]([C@@H]2CC1)C)O)C | 21 |
| 3ihz_ligand_1_4.mol2 | 3ihz | 0.547619 | -6.66 | [C@@H]1(CC[C@@H](CC1)O)/C=C\C | 10 |
| 1fkf_ligand_1_3.mol2 | 1fkf | 0.547619 | -6.43 | [C@@H]1(CC[C@@H](CC1)O)/C=C\C | 10 |
| 1a7x_ligand_1_5.mol2 | 1a7x | 0.547619 | -5.97 | [C@@H]1(CC[C@@H](CC1)O)/C=C\C | 10 |
102 ,
11

