
Common name
(1R,2R,3aS,9aS)-1-methyl-2,3,3a,4,9,9a-hexahydro-1H-cyclopenta[b]naphthalen-2-ol
IUPAC name
(1R,2R,3aS,9aS)-1-methyl-2,3,3a,4,9,9a-hexahydro-1H-cyclopenta[b]naphthalen-2-ol
SMILES
C12C(CC(C1C)O)Cc3c(cccc3)C2
Common name
(1R,2R,3aS,9aS)-1-methyl-2,3,3a,4,9,9a-hexahydro-1H-cyclopenta[b]naphthalen-2-ol
IUPAC name
(1R,2R,3aS,9aS)-1-methyl-2,3,3a,4,9,9a-hexahydro-1H-cyclopenta[b]naphthalen-2-ol
SMILES
C12C(CC(C1C)O)Cc3c(cccc3)C2
INCHI
InChI=1S/C14H18O/c1-9-13-7-11-5-3-2-4-10(11)6-12(13)8-14(9)15/h2-5,9,12-15H,6-8H2,1H3/t9-,12+,13-,14-/m1/s1
FORMULA
C14H18O

Common name
(1R,2R,3aS,9aS)-1-methyl-2,3,3a,4,9,9a-hexahydro-1H-cyclopenta[b]naphthalen-2-ol
IUPAC name
(1R,2R,3aS,9aS)-1-methyl-2,3,3a,4,9,9a-hexahydro-1H-cyclopenta[b]naphthalen-2-ol
Molecular weight
202.292
clogP
2.821
clogS
-2.626
Frequency
0.0003
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
20.23
Number of Rings
3
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00256 | Treprostinil |
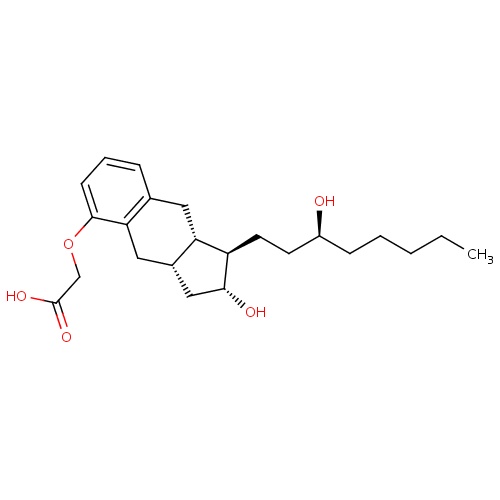
|
Antithrombins; Anticoagulants; Antihypertensive Agents; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; | For use as a continuous subcutaneous infusion or intravenous infusion (for those not able to tolerate a subcutaneous infusion) for the treatment of pulmonary arterial hypertension in patients with NYHA Class II-IV symptoms to diminish symptoms associated with exercise. |
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2b1z_ligand.mol2 | 2b1z | 0.823529 | -10.14 | C[C@]12CCc3c4ccc(O)cc4ccc3[C@@H]1CC[C@@]2(C)O | 22 |
| 4n8d_ligand_2_1.mol2 | 4n8d | 0.809524 | -7.29 | [C@@H]1(CC[C@@H](CC1)O)c1ccccc1 | 13 |
| 1lhu_ligand.mol2 | 1lhu | 0.792453 | -10.22 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |
| 1g50_ligand.mol2 | 1g50 | 0.792453 | -10.01 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |
| 1jgl_ligand.mol2 | 1jgl | 0.792453 | -9.99 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |
| 2j7x_ligand.mol2 | 2j7x | 0.792453 | -9.96 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |
| 1a52_ligand.mol2 | 1a52 | 0.792453 | -9.84 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |
| 1qku_ligand.mol2 | 1qku | 0.792453 | -9.81 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |
| 1qkt_ligand.mol2 | 1qkt | 0.792453 | -9.64 | c1cc(O)cc2CC[C@@H]3[C@@H](c12)CC[C@]1([C@H]3CC[C@@H]1O)C | 21 |

