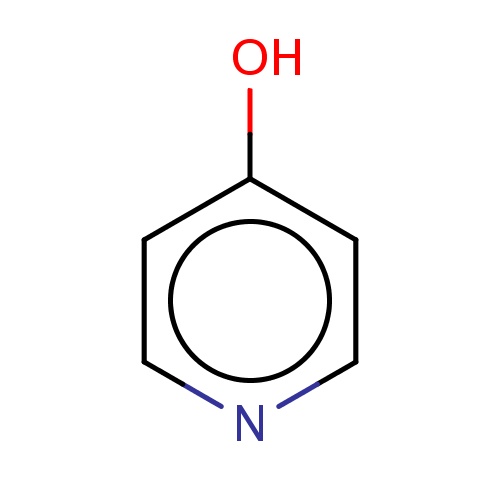
Common name
pyridin-4-ol
IUPAC name
pyridin-4-ol
SMILES
Oc1ccncc1
Common name
pyridin-4-ol
IUPAC name
pyridin-4-ol
SMILES
Oc1ccncc1
INCHI
InChI=1S/C5H5NO/c7-5-1-3-6-4-2-5/h1-4H,(H,6,7)
FORMULA
C5H5NO

Common name
pyridin-4-ol
IUPAC name
pyridin-4-ol
Molecular weight
95.099
clogP
1.026
clogS
-0.756
Frequency
0.0007
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
33.12
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00278 | Sorafenib |
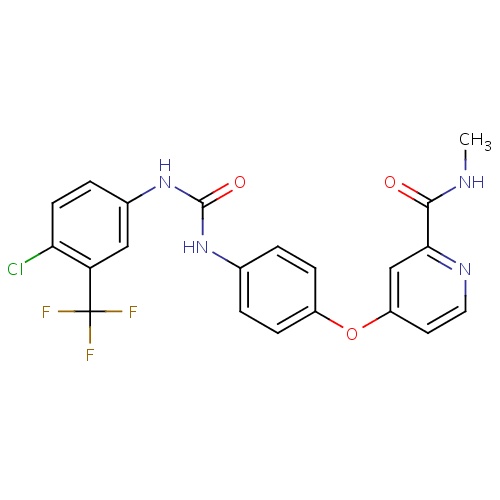
|
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Sorafenib is indicated for the treatment of unresectable hepatocellular carcinoma and advanced renal cell carcinoma. |
| FDBD01561 | Regorafenib |
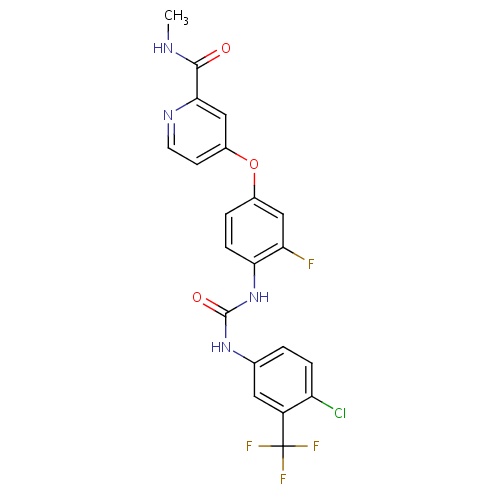
|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Regorafenib is indicated for the treatment of patients with metastatic colorectal cancer (CRC) who have been previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy, an anti-VEGF therapy, and, if KRAS wild type, an anti-EGFR therapy. Regorafenib is also indicated for the treatment of patients with locally advanced, unresectable or metastatic gastrointestinal stromal tumor (GIST) who have been previously treated with imatinib mesylate and sunitinib malate. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 5ct7_ligand_1_1.mol2 | 5ct7 | 1 | -6.23 | c1c(ccnc1)O | 7 |
| 2qu5_ligand_1_1.mol2 | 2qu5 | 1 | -6.22 | c1cnccc1O | 7 |
| 2qu6_ligand_1_3.mol2 | 2qu6 | 1 | -6.19 | c1nccc(c1)O | 7 |
| 5ar7_ligand_1_3.mol2 | 5ar7 | 1 | -6.18 | Oc1ccncc1 | 7 |
| 1uwh_ligand_1_3.mol2 | 1uwh | 1 | -6.15 | Oc1ccncc1 | 7 |
| 2wot_ligand_1_3.mol2 | 2wot | 1 | -6.12 | c1cc(ccn1)O | 7 |
| 2wou_ligand_1_1.mol2 | 2wou | 1 | -6.10 | Oc1ccncc1 | 7 |
665 ,
67

