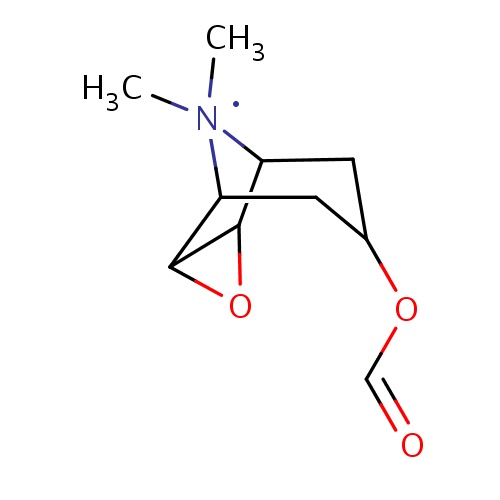
Common name
(dimethylBLAHyl) formate
IUPAC name
(dimethylBLAHyl) formate
SMILES
C12C(O1)C3[N](C2CC(C3)OC=O)(C)C
Common name
(dimethylBLAHyl) formate
IUPAC name
(dimethylBLAHyl) formate
SMILES
C12C(O1)C3[N](C2CC(C3)OC=O)(C)C
INCHI
InChI=1S/C10H16NO3/c1-11(2)7-3-6(13-5-12)4-8(11)10-9(7)14-10/h5-10H,3-4H2,1-2H3/t6-,7-,8+,9-,10+
FORMULA
C10H16NO3

Common name
(dimethylBLAHyl) formate
IUPAC name
(dimethylBLAHyl) formate
Molecular weight
198.239
clogP
-2.188
clogS
-0.923
Frequency
0.0003
HBond Acceptor
3
HBond Donor
0
Total PolarSurface Area
38.83
Number of Rings
3
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
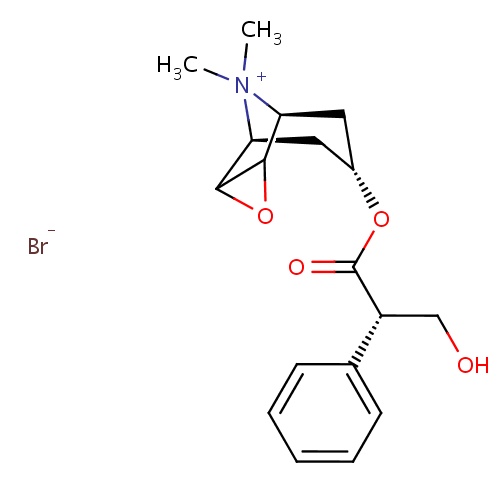
| FDBD00337 | Methylscopolamine bromide |
|
Parasympatholytics; Cholinergic Antagonists; Antispasmodics; | Used as adjunctive therapy for the treatment of peptic ulcer. Also used to treat nausea and vomiting due to motion sickness. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2dxs_ligand_frag_2.mol2 | 2dxs | 0.761194 | -7.81 | C1CC[C@H]2[C@@H]([N@@H+]3[C@H]([C@H]4[C@H](CCCC4)OCC3)C2)C1 | 18 |
| 1i7z_ligand_frag_1.mol2 | 1i7z | 0.71875 | -7.16 | C1[C@H](C[C@H]2[N@@H+]([C@@H]1CC2)C)OC=O | 12 |
| 2pgz_ligand_frag_1.mol2 | 2pgz | 0.71875 | -7.02 | C1[C@@H]2[N@H+]([C@@H](CC2)C[C@@H]1OC=O)C | 12 |
| 1q72_ligand_frag_1.mol2 | 1q72 | 0.71875 | -6.48 | C1[C@@H]2[N@H+]([C@@H](CC2)C[C@@H]1OC=O)C | 12 |
| 2qp8_ligand_4_708.mol2 | 2qp8 | 0.707692 | -6.80 | C(C)O[C@@H]1C[C@@H]([NH2+]CC1)[C@@H](O)C | 12 |
| 2qmd_ligand_5_720.mol2 | 2qmd | 0.681818 | -6.58 | [C@H]1([NH2+]C[C@@H](C1)OC)[C@H](CC)O | 11 |
| 2arm_ligand_1_0.mol2 | 2arm | 0.680556 | -6.16 | C[N@@H+]1[C@@]23CC[C@@]12C[C@H](C3)OC(=O)C | 13 |
| 2dxs_ligand_1_1.mol2 | 2dxs | 0.675325 | -7.86 | NC(=O)[C@@H]1CC[C@H]2[C@@H]([N@@H+]3[C@H]([C@H]4[C@H](CCCC4)OCC3)C2)C1 | 21 |
| 2qp8_ligand_3_284.mol2 | 2qp8 | 0.661538 | -6.42 | C(C)O[C@@H]1C[C@@H]([NH2+]CC1)CO | 11 |
104 ,
11

